Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Binding energy of a certain nucleus is $$18 \times 10^8 \text{ J}$$. How much is the difference between total mass of all the nucleons and nuclear mass of the given nucleus:
Binding energy = $$18 \times 10^8$$ J. Find the mass difference between nucleons and the nucleus.
$$E = \Delta m \cdot c^2$$
The binding energy represents the energy equivalent of the mass defect $$\Delta m$$.
$$\Delta m = \frac{E}{c^2} = \frac{18 \times 10^8}{(3 \times 10^8)^2} = \frac{18 \times 10^8}{9 \times 10^{16}} = 2 \times 10^{-8} \text{ kg}$$
$$1 \text{ kg} = 10^9 \; \mu\text{g}$$, so:
$$\Delta m = 2 \times 10^{-8} \times 10^9 = 20 \; \mu\text{g}$$
The correct answer is Option (2): 20 $$\mu$$g.
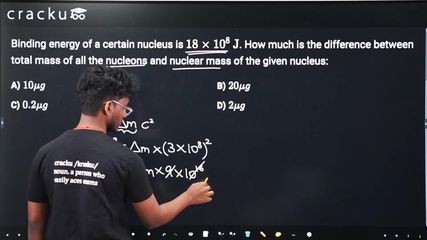
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
