Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by:
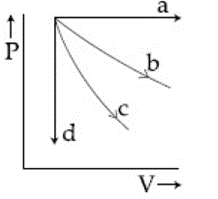
Based on the principles of thermodynamics and the slopes of the curves on a $$P-V$$ diagram, the processes are assigned as follows:
Correct Assignment:
Final Order:
$$\boxed{d, a, b, c}$$
Create a FREE account and get:
Educational materials for JEE preparation
