Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
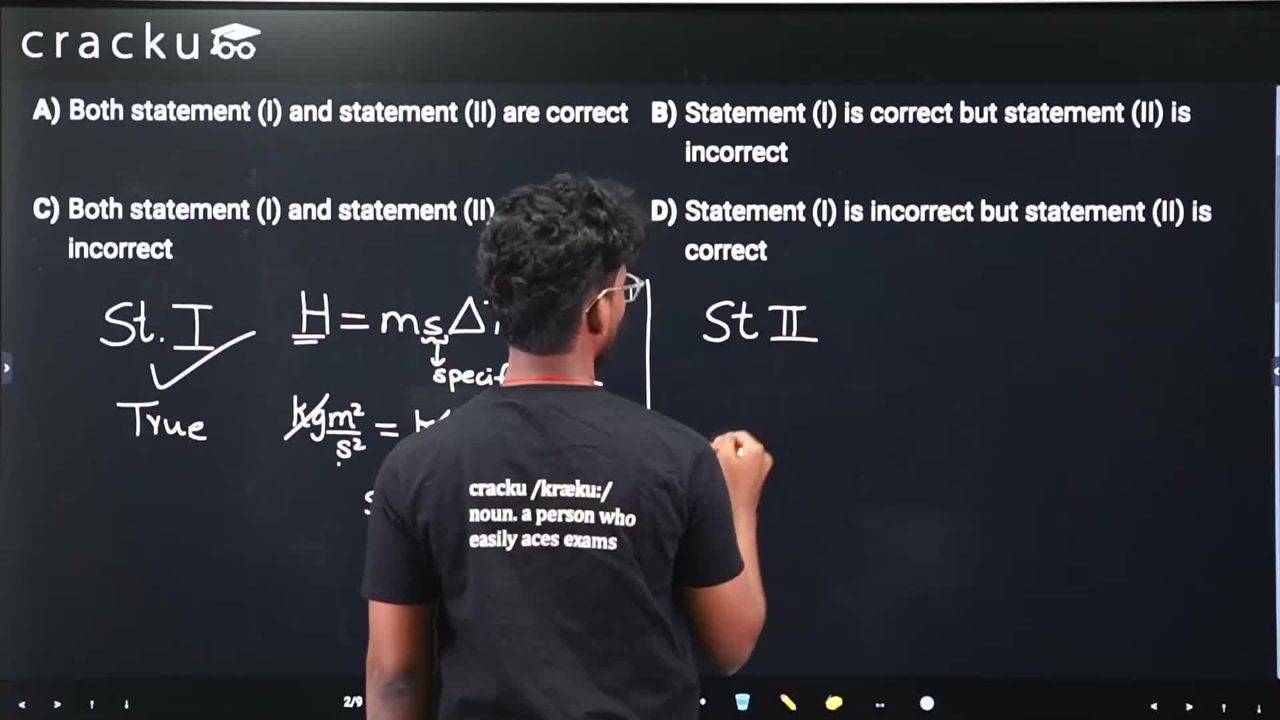
Given below are two statements : Statement (I) : Dimensions of specific heat is $$[L^2 T^{-2} K^{-1}]$$. Statement (II) : Dimensions of gas constant is $$[ML^2 T^{-1} K^{-1}]$$. In the light of the above statements, choose the most appropriate answer from the options given below.
We need to evaluate two statements about dimensions.
Statement I: Dimensions of specific heat is $$[L^2 T^{-2} K^{-1}]$$.
Specific heat = Energy / (mass × temperature).
Dimensions: $$\frac{[ML^2T^{-2}]}{[M][K]} = [L^2T^{-2}K^{-1}]$$
Statement I is correct. ✓
Statement II: Dimensions of gas constant is $$[ML^2T^{-1}K^{-1}]$$.
Gas constant R = Energy / (mol × temperature) (from PV = nRT).
Dimensions: $$\frac{[ML^2T^{-2}]}{[\text{mol}][K]} = [ML^2T^{-2}K^{-1}\text{mol}^{-1}]$$
The given dimension has $$T^{-1}$$ instead of $$T^{-2}$$. Statement II is incorrect. ✗
The correct answer is Option 2: Statement (I) is correct but Statement (II) is incorrect.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
