NTA JEE Mains 9th April 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 31
The candela is the luminous intensity, in a given direction, of a source that emits monochromatic radiation of frequency '$$A$$' $$\times 10^{12}$$ hertz and that has a radiant intensity in that direction of $$\frac{1}{B'}$$ watt per steradian. '$$A$$' and '$$B$$' are respectively
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 32
The electronic configuration of Einsteinium is : (Given atomic number of Einsteinium $$= 99$$)
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 33
Match List I with List II:
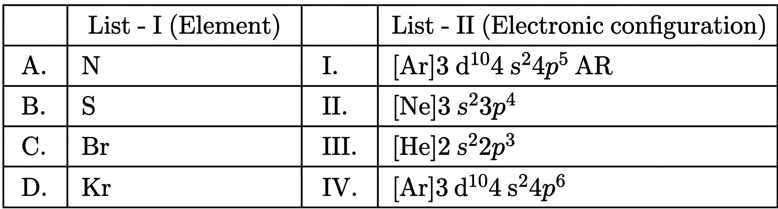
Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 34
Match List I with List II:
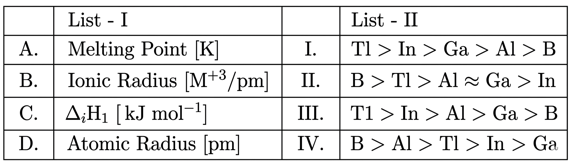
Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 35
The correct increasing order for bond angles among $$BF_3$$, $$PF_3$$ and $$ClF_3$$ is :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 36
The incorrect statement regarding ethyne is
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 37
For a sparingly soluble salt $$AB_2$$, the equilibrium concentrations of $$A^{2+}$$ ions and $$B^-$$ ions are $$1.2 \times 10^{-4} M$$ and $$0.24 \times 10^{-3} M$$, respectively. The solubility product of $$AB_2$$ is :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 38
The correct stability order of the following resonance structures of $$CH_3 - CH = CH - CHO$$ i
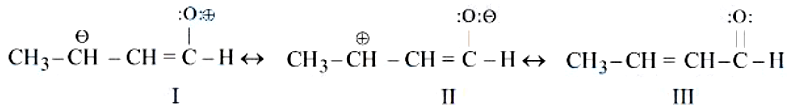
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 39
Total number of stereo isomers possible for the given structure :
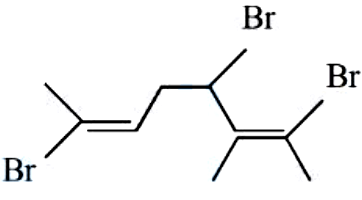
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 40
Which out of the following is a correct equation to show change in molar conductivity with respect to concentration for a weak electrolyte, if the symbols carry their usual meaning :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 41
Match List I with List II:

Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 42
Given below are two statements: Statement I : The higher oxidation states are more stable down the group among transition elements unlike p-block elements. Statement II : Copper can not liberate hydrogen from weak acids. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 43
Match List I with List II:
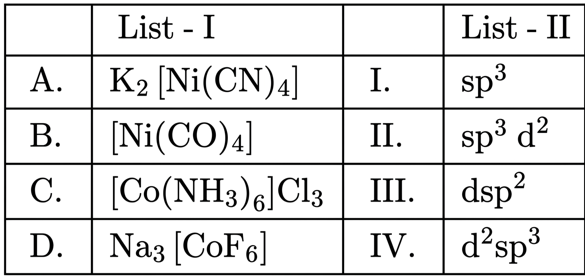
Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 44
The coordination environment of $$Ca^{2+}$$ ion in its complex with $$EDTA^{4-}$$ is :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 45
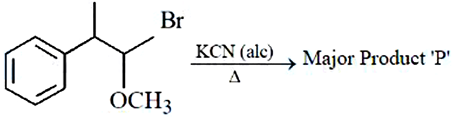
In the above reaction product '$$P$$' is :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 46
Match List I with List II:
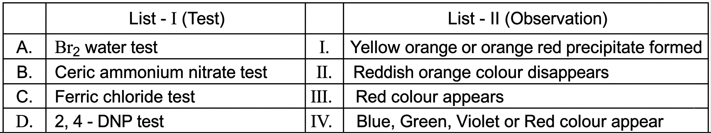
Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 47
Which of the following compound can give positive iodoform test when treated with aqueous $$KOH$$ solution followed by potassium hypoiodite.
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 48
Which of the following compounds will give silver mirror with ammoniacal silver nitrate? A. Formic acid B. Formaldehyde C. Benzaldehyde D. Acetone. Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 49
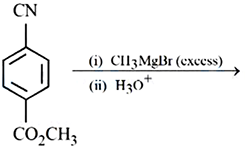
Major product of the following reaction is: A compound with $$CO_2CH_3$$ group reacts with (i) $$CH_3MgBr$$ (excess) (ii) $$H_3O^+$$
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 50
The incorrect statement about Glucose is :
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 51
Based on Heisenberg's uncertainty principle, the uncertainty in the velocity of the electron to be found within an atomic nucleus of diameter $$10^{-15} \text{ m}$$ is ______ $$\times 10^9 \text{ ms}^{-1}$$ (nearest integer) [Given : mass of electron $$= 9.1 \times 10^{-31} \text{ kg}$$, Planck's constant $$(h) = 6.626 \times 10^{-34} \text{ Js}$$] (Value of $$\pi = 3.14$$)
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 52
Total number of electrons present in $$(\pi^*)$$ molecular orbitals of $$O_2$$, $$O_2^+$$ and $$O_2^-$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 53
When $$\Delta H_{vap} = 30 \text{ kJ/mol}$$ and $$\Delta S_{vap} = 75 \text{ J mol}^{-1} \text{K}^{-1}$$, then the temperature of vapour, at one atmosphere is ______ K.
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 54
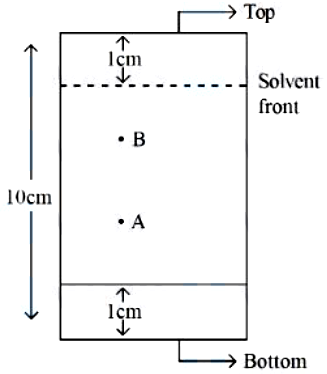
In the given TLC, the distance of spot A & B are $$5 \text{ cm}$$ & $$7 \text{ cm}$$, from the bottom of TLC plate, respectively. $$R_f$$ value of B is $$x \times 10^{-1}$$ times more than A. The value of $$x$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 55
Number of compounds from the following which cannot undergo Friedel-Crafts reactions is: ________. toluene, nitrobenzene, xylene, cumene, aniline, chlorobenzene, $$m$$-nitroaniline, $$m$$-dinitrobenzene
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 56
The vapour pressure of pure benzene and methyl benzene at $$27°C$$ is given as 80 Torr and 24 Torr, respectively. The mole fraction of methyl benzene in vapour phase, in equilibrium with an equimolar mixture of those two liquids (ideal solution) at the same temperature is ______ $$\times 10^{-2}$$ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 57
Consider the following first order gas phase reaction at constant temperature $$A(g) \to 2B(g) + C(g)$$. If the total pressure of the gases is found to be 200 torr after 23 sec. and 300 torr upon the complete decomposition of A after a very long time, then the rate constant of the given reaction is ______ $$\times 10^{-2} \text{ s}^{-1}$$ (nearest integer) [Given : $$\log_{10}(2) = 0.301$$]
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 58
Number of oxygen atoms present in chemical formula of fuming sulphuric acid is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 59
A transition metal '$$M$$' among $$Sc, Ti, V, Cr, Mn$$ and $$Fe$$ has the highest second ionisation enthalpy. The spin-only magnetic moment value of $$M^+$$ ion is ______ BM (Near integer) (Given atomic number $$Sc : 21, Ti : 22, V : 23, Cr : 24, Mn : 25, Fe : 26$$)
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 2 - Chemistry - Question 60
$$M^{2+} + H_2S \to A \text{ (Black precipitate)} + \text{by product}$$
$$A + \text{aqua regia} \to B + NOCl + S + H_2O$$
$$B + KNO_2 + CH_3COOH \to C + \text{by product}$$
Consider the following test for a group-IV cation. The spin-only magnetic moment value of the metal complex C is ______ BM (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




