NTA JEE Mains 9th April 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 31
Compare the energies of following sets of quantum numbers for multielectron system. (A) $$n = 4, l = 1$$ (B) $$n = 4, l = 2$$ (C) $$n = 3, l = 1$$ (D) $$n = 3, l = 2$$ (E) $$n = 4, l = 0$$. Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 32
Given below are two statements : Statement (I) : The oxidation state of an element in a particular compound is the charge acquired by its atom on the basis of electron gain enthalpy consideration from other atoms in the molecule. Statement (II) : $$p\pi - p\pi$$ bond formation is more prevalent in second period elements over other periods. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 33
In which one of the following pairs the central atoms exhibit $$sp^2$$ hybridization ?
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 34
Identify the incorrect statements regarding primary standard of titrimetric analysis. (A) It should be purely available in dry form. (B) It should not undergo chemical change in air. (C) It should be hygroscopic and should react with another chemical instantaneously and stoichiometrically. (D) It should be readily soluble in water. (E) $$KMnO_4$$ & $$NaOH$$ can be used as primary standard. Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 35
Methods used for purification of organic compounds are based on :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 36
Correct order of basic strength of Pyrrole,
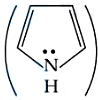
Pyridine,
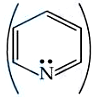
and Piperidine
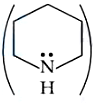
is:
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 37
For the given compounds, the correct order of increasing $$pK_a$$ value :
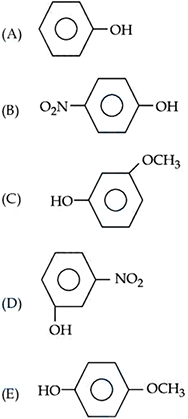
Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 38
Relative stability of the contributing structures is
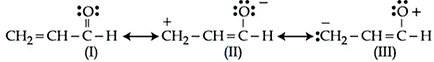
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 39
Identify the product A and product B in the following set of reactions.
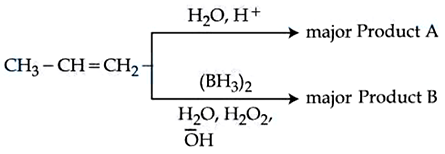
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 40
The molar conductivity for electrolytes A and B are plotted against $$C^{1/2}$$ as shown below. Electrolytes A and B respectively are
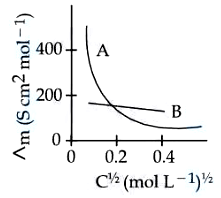
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 41
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : Both rhombic and monoclinic sulphur exist as $$S_8$$ while oxygen exists as $$O_2$$. Reason (R) : Oxygen forms $$p\pi - p\pi$$ multiple bonds with itself and other elements having small size and high electronegativity like C, N, which is not possible for sulphur. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 42
On reaction of Lead Sulphide with dilute nitric acid which of the following is not formed?
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 43
$$0.05$$ M $$CuSO_4$$ when treated with $$0.01$$ M $$K_2Cr_2O_7$$ gives green colour solution of $$Cu_2Cr_2O_7$$.
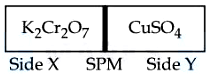
The two solutions are separated as shown below : [SPM : Semi Permeable Membrane]. Due to osmosis :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 44
Electronic configuration of Cu(II) is $$3d^9$$ whereas that of Cu(I) is $$3d^{10}$$. Which of the following is correct?
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 45
The $$F^-$$ ions make the enamel on teeth much harder by converting hydroxyapatite (the enamel on the surface of teeth) into much harder fluoroapatite having the formula.
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 46
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : The total number of geometrical isomers shown by $$[Co(en)_2Cl_2]^+$$ complex ion is three. Reason (R) : $$[Co(en)_2Cl_2]^+$$ complex ion has an octahedral geometry. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 47
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : $$S_N2$$ reaction of $$C_6H_5CH_2Br$$ occurs more readily than the $$S_N2$$ reaction of $$CH_3CH_2Br$$. Reason (R) : The partially bonded unhybridized p-orbital that develops in the trigonal bipyramidal transition state is stabilized by conjugation with the phenyl ring. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 48
In the following sequence of reaction, the major products B and C respectively are :
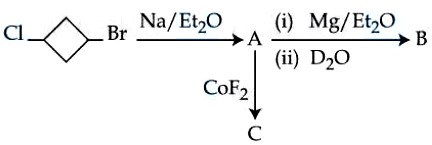
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 49
Identify major product "X" formed in the following reaction :
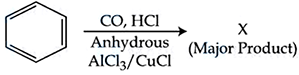
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 50
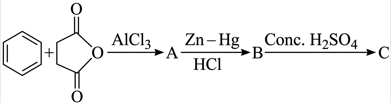
What is the structure of C
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 51
Molarity (M) of an aqueous solution containing $$x$$ g of anhyd. $$CuSO_4$$ in $$500$$ mL solution at $$32°C$$ is $$2 \times 10^{-1}$$ M. Its molality will be ______ $$\times 10^{-3}$$ m. (nearest integer). [Given density of the solution $$= 1.25$$ g/mL]
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 52
The total number of species from the following in which one unpaired electron is present, is _______
$$N_2, O_2, C_2^-, O_2^-, O_2^{2-}, H_2^+, CN^-, He_2^+$$
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 53
When equal volume of $$1$$ M HCl and $$1$$ M $$H_2SO_4$$ are separately neutralised by excess volume of $$1$$ M NaOH solution. $$x$$ and $$y$$ kJ of heat is liberated respectively. The value of $$y/x$$ is _______
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 54
The heat of solution of anhydrous $$CuSO_4$$ and $$CuSO_4 \cdot 5H_2O$$ are $$-70$$ kJ mol$$^{-1}$$ and $$+12$$ kJ mol$$^{-1}$$ respectively. The heat of hydration of $$CuSO_4$$ to $$CuSO_4 \cdot 5H_2O$$ is $$-x$$ kJ. The value of $$x$$ is ______ (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 55
How many compounds among the following compounds show inductive, mesomeric as well as hyperconjugation effects?
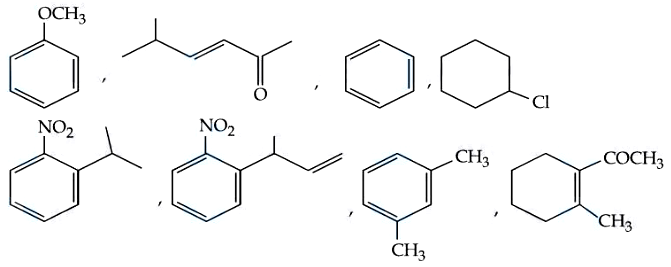
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 56
The standard reduction potentials at $$298$$ K for the following half cells are given below :
$$Cr_2O_7^{2-} + 14H^+ + 6e^- \rightarrow 2Cr^{3+} + 7H_2O$$, $$E° = 1.33$$ V
$$Fe^{3+}(aq) + 3e^- \rightarrow Fe$$, $$E° = -0.04$$ V
$$Ni^{2+}(aq) + 2e^- \rightarrow Ni$$, $$E° = -0.25$$ V
$$Ag^+(aq) + e^- \rightarrow Ag$$, $$E° = 0.80$$ V
$$Au^{3+}(aq) + 3e^- \rightarrow Au$$, $$E° = 1.40$$ V
Consider the given electrochemical reactions, The number of metal(s) which will be oxidized by $$Cr_2O_7^{2-}$$, in aqueous solution is ______
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 57
Given below are two statements : Statement I: The rate law for the reaction $$A + B \rightarrow C$$ is rate $$(r) = k[A]^2[B]$$. When the concentration of both A and B is doubled, the reaction rate is increased "$$x$$" times. Statement II :
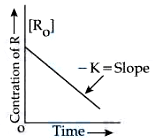
The figure is showing "the variation in concentration against time plot" for a "$$y$$" order reaction. The Value of $$x + y$$ is ______
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 58
Number of colourless lanthanoid ions among the following is ________
$$Eu^{3+}, Lu^{3+}, Nd^{3+}, La^{3+}, Sm^{3+}$$
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 59
Number of ambidentate ligands among the following is ______
$$NO_2^-, SCN^-, C_2O_4^{2-}, NH_3, CN^-, SO_4^{2-}, H_2O$$
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Chemistry - Question 60
Total number of essential amino acid among the given list of amino acids is _______ Arginine, Phenylalanine, Aspartic acid, Cysteine, Histidine, Valine, Proline
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




