NTA JEE Mains 8th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 8th April 2024 Shift 2 - Question 51
Wavenumber for a radiation having wavelength $$5800 \text{ Å}$$ is $$x \times 10 \text{ cm}^{-1}$$. The value of $$x$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 52
Number of molecules having bond order 2 from the following molecules is _____ : $$C_2, O_2, Be_2, Li_2, Ne_2, N_2, He_2$$
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 53
$$\Delta_{vap}H^{\ominus}$$ for water is $$+40.79 \text{ kJ mol}^{-1}$$ at 1 bar and $$100°C$$. Change in internal energy for this vapourisation under same condition is _____ $$\text{kJ mol}^{-1}$$. (Given $$R = 8.3 \text{ JK}^{-1}\text{mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 54
Total number of optically active compounds from the following is ____
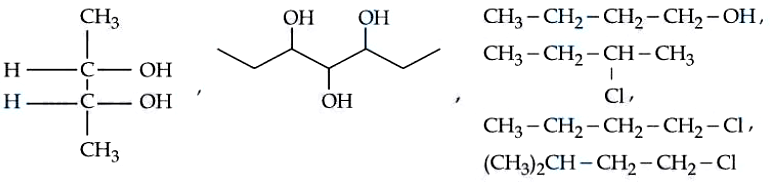
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 55
Total number of aromatic compounds among the following compounds is _____
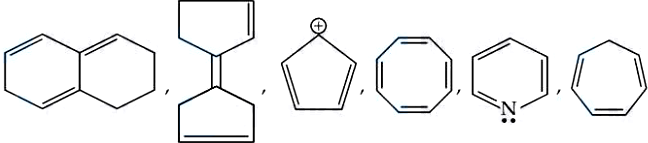
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 56
A solution is prepared by adding 1 mole ethyl alcohol in 9 mole water. The mass percent of solute in the solution is _____ (Given : Molar mass in g/mol: Ethyl alcohol : 46, water: 18)
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 57
Molality of an aqueous solution of urea is 4.44 m. Mole fraction of urea in solution is $$x \times 10^{-3}$$. Value of $$x$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 58
Total number of unpaired electrons in the complex ions $$[Co(NH_3)_6]^{3+}$$ and $$[NiCl_4]^{2-}$$ is _____
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 59
Two moles of benzaldehyde and one mole of acetone under alkaline conditions using aqueous NaOH after heating gives $$x$$ as the major product. The number of $$\pi$$ bonds in the product $$x$$ is _____
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Question 60
The total number of carbon atoms present in tyrosine, an amino acid, is _____
789
456
123
0.-
Clear All


