NTA JEE Mains 6th April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 6th April Shift 2 2026 - Question 61
Which of the following sequences of hybridisation, geometry and magnetic nature are correct for the given coordination compounds?
A. $$[NiCl_4]^{2-}$$ -- sp$$^3$$, tetrahedral, paramagnetic
B. $$[Ni(NH_3)_6]^{2+}$$ -- sp$$^3$$d$$^2$$, octahedral, paramagnetic
C. $$[Ni(CO)_4]$$ -- sp$$^3$$, tetrahedral, paramagnetic
D. $$[Ni(CN)_4]^{2-}$$ -- dsp$$^2$$, square planar, diamagnetic
Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 62
Given below are two statements :
Statement I : A mixture of C$$_{12}$$H$$_{22}$$O$$_{11}$$ (sugar) and NaCl can be separated by dissolving sugar in alcohol, due to differential solubility.
Statement II : Rose essence from rose petals is separated by steam distillation due to its high volatility and insolubility in H$$_2$$O.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 63
Shown below is the structure of methyl acetate with three different $$\alpha$$, $$\beta$$ and $$\gamma$$ carbon-oxygen bonds.
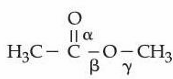
The correct order of bond lengths of these bonds is :
NTA JEE Mains 6th April Shift 2 2026 - Question 64
'x' is the product which is obtained by the hydrolysis of prop-1-yne in the presence of mercuric sulphate under dilute acidic medium at 333 K. 'y' is the product which is obtained by the reaction of ethane nitrile with methyl magnesium bromide in dry ether followed by hydrolysis. IUPAC name of product obtained from 'x' and 'y' in the presence of barium hydroxide followed by heating is :
NTA JEE Mains 6th April Shift 2 2026 - Question 65
An optically active alkyl bromide C$$_4$$H$$_9$$Br, reacts with ethanolic KOH to form major compound [A] which reacts with bromine to give compound [B]. Compound [B] reacts with ethanolic KOH and sodamide to give compound [C]. One molecule of water adds to compound [C] on warming with mercuric sulphate and dilute sulphuric acid at 333 K to form compound [D]. The functional group in compound D will be confirmed by :
NTA JEE Mains 6th April Shift 2 2026 - Question 66
Consider the following reaction.
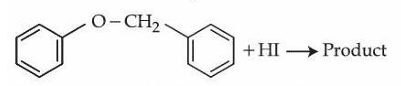
Statement I : In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
Statement II : In the above reaction, the $$-O-CH_2-$$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 67
Consider the following organic reaction sequence. Choose the final product (X) from the following (consider the major product in all intermediate reactions)
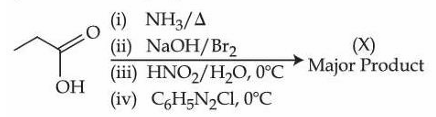
NTA JEE Mains 6th April Shift 2 2026 - Question 68
The number of compounds from the following which can undergo reaction with Br$$_2$$/KOH (alcoholic) to give respective products and these respective products can also be obtained separately by Gabriel phthalimide reaction is :
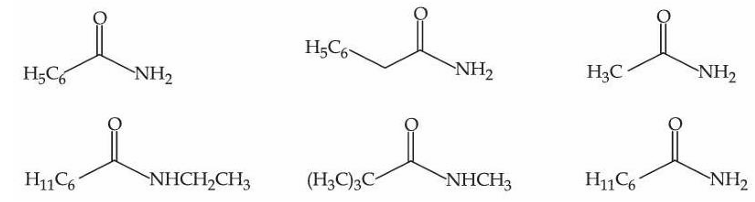
NTA JEE Mains 6th April Shift 2 2026 - Question 69
Consider the following reactions. Total number of electrons in the $$\pi$$ bonds and lone pair of electrons in the product (X) is :

NTA JEE Mains 6th April Shift 2 2026 - Question 70
Treatment of a gas 'X' with a freshly prepared ferrous sulphate solution gives a compound 'Y' as a brown ring. The compounds X and Y are.


