NTA JEE Mains 6th April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 6th April Shift 2 2026 - Question 51
Which of the following contain the same number of atoms?
(Given : Molar mass in g mol$$^{-1}$$ of H, He, O and S are 1, 4, 16 and 32 respectively)
A. 2 g of O$$_2$$ gas
B. 4 g of SO$$_2$$ gas
C. 1400 mL of O$$_2$$ at STP
D. 0.05 L of He at STP
E. 0.0625 mol of H$$_2$$ gas
Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 52
The Bohr radius of a hydrogen like species is 70.53 pm. The species and the stationary state (n) are respectively (Given : Hydrogen atom Bohr radius is 52.9 pm)
NTA JEE Mains 6th April Shift 2 2026 - Question 53
Given below are two statements :
Statement I : The number of compounds among SO$$_2$$, SO$$_3$$, SF$$_4$$, SF$$_6$$ and H$$_2$$S in which sulphur does not obey the Octet rule is 3.
Statement II : Among [H$$_2$$O, ClF$$_3$$, SF$$_4$$], [NH$$_3$$, BrF$$_5$$, SF$$_4$$], [BrF$$_5$$, ClF$$_3$$, XeF$$_4$$] and [XeF$$_4$$, ClF$$_3$$, H$$_2$$O], the number of sets in which all the molecules have one lone pair of electrons on the central atom is 1.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 54
Match List - I with List - II.
Given $$V_1$$ and $$V_2$$ are initial and final volumes respectively.

Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 55
Given below are two statements :
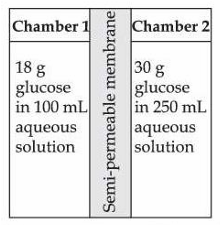
Statement I : H$$_2$$O molecules move from the chamber 1 to chamber 2.
Statement II : The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass = 174 g/mol) in 2 L of water (at 27 °C) is 0.0107 bar. (Given: R = 0.083 dm$$^3$$ bar K$$^{-1}$$ mol$$^{-1}$$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 56
Given is a concentrated solution of a weak electrolyte $$A_xB_y$$ of concentration 'c' and dissociation constant 'K'. The degree of dissociation is given by :
NTA JEE Mains 6th April Shift 2 2026 - Question 57
For a general redox reaction
Anode: $$\text{Red}_1 \to \text{Ox}_1^{n_1+} + n_1 e^-$$
Cathode: $$\text{Ox}_2 + n_2 e^- \to \text{Red}_2^{n_2-}$$
Which of the following statement is **incorrect** ?
NTA JEE Mains 6th April Shift 2 2026 - Question 58
In a period, the first ionisation enthalpy of the element at extreme left and the negative electron gain enthalpy of the extreme right element, except noble gases, are respectively.
NTA JEE Mains 6th April Shift 2 2026 - Question 59
Given below are two statements :
Statement I : $$F_2O < H_2O < Cl_2O$$ is the correct trend in terms of bond angle.
Statement II : SiF$$_4$$, SnF$$_4$$ and PbF$$_4$$ are ionic in nature.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Question 60
The correct order of first ($$\Delta_i H_1$$) and second ($$\Delta_i H_2$$) ionisation enthalpy values of Cr and Mn are :
A. $$\Delta_i H_1$$ : Cr > Mn
B. $$\Delta_i H_2$$ : Cr > Mn
C. $$\Delta_i H_1$$ : Mn > Cr
D. $$\Delta_i H_2$$ : Mn > Cr
Choose the correct answer from the options given below :


