NTA JEE Mains 6th April Shift 1 2026
For the following questions answer them individually
NTA JEE Mains 6th April Shift 1 2026 - Question 71
First and second ionization enthalpies of lithium are 520 kJ mol$$^{-1}$$ and 7297 kJ mol$$^{-1}$$ respectively. Energy required to convert 3.5 mg lithium (g) into Li$$^{2+}$$(g) [Li(g) $$\to$$ Li$$^{2+}$$(g)] is _______ kJ mol$$^{-1}$$. (nearest integer)
[Molar mass of Li = 7 g mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Question 72
Consider the following sequence of reactions.
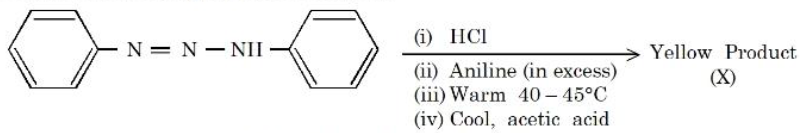
The percentage of nitrogen in the yellow product (X) formed is _______ %. (Nearest Integer)
(Given Molar mass in g mol$$^{-1}$$ H:1, C:12, N:14)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Question 73
4.7 g of phenol is heated with Zn to give product X. If this reaction goes to 60% completion then the number of moles of compound X formed will be _______ $$\times 10^{-2}$$. (Nearest Integer)
(Given molar mass in g mol$$^{-1}$$: H:1, C:12, O:16)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Question 74
Sucrose hydrolyses in acidic medium into glucose and fructose by first order rate law with $$t_{1/2} = 3$$ hour. The percentage of sucrose remaining after 6 hours is _______. (Nearest integer)
(Given: log 2 = 0.3010 and log 3 = 0.4771)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Question 75
Consider the reaction X $$\rightleftharpoons$$ Y at 300 K. If $$\Delta H^\theta$$ and K are 28.40 kJ mol$$^{-1}$$ and $$1.8 \times 10^{-7}$$ at the same temperature, then the magnitude of $$\Delta S^\theta$$ for the reaction in J K$$^{-1}$$ mol$$^{-1}$$ is _______. (Nearest integer)
(Given: R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$, ln 10 = 2.3, log 3 = 0.48, log 2 = 0.30)
789
456
123
0.-
Clear All


