NTA JEE Mains 6th April Shift 1 2026
For the following questions answer them individually
NTA JEE Mains 6th April Shift 1 2026 - Question 61
Match the LIST-I with LIST-II
| List-I Electronic configuration of tetrahedral metal ion | List-II Crystal Field Stabilization Energy ($$\Delta_t$$) | ||
|---|---|---|---|
| A. | d$$^2$$ | I. | -0.6 |
| B. | d$$^4$$ | II. | -0.8 |
| C. | d$$^6$$ | III. | -1.2 |
| D. | d$$^8$$ | IV. | -0.4 |
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Question 62
Which of the following are true about the energy of the given d-orbitals of a tetrahedral complex?
A. $$d_{xy} = d_{xz} > d_{x^2-y^2}$$
B. $$d_{xy} = d_{yz} > d_{z^2}$$
C. $$d_{x^2-y^2} > d_{z^2} > d_{xz}$$
D. $$d_{x^2-y^2} = d_{z^2} < d_{xz}$$
Choose the correct answer from the given below:
NTA JEE Mains 6th April Shift 1 2026 - Question 63
R$$_f$$ value for 2-methylpropene in a solvent system (Ethyl acetate + ether) is 0.42. 2-methylpropene is treated with dilute H$$_2$$SO$$_4$$ to give major organic product (X). R$$_f$$ value for (X) in the same solvent system under identical condition will be:
NTA JEE Mains 6th April Shift 1 2026 - Question 64
Given below are two statements:
Statement I: 2,6-diethylcyclohexanone and 6-methyl-2-n-propylcyclohexanone are metamers.
Statement II: 2,2,6,6-tetramethylcyclohexanone exhibits keto-enol tautomerism.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Question 65
Given below are two statements:
Statement I: Methane can be prepared by decarboxylation of sodium ethanoate, Kolbe's electrolysis of sodium acetate and reaction of CH$$_3$$MgBr with water.
Statement II: Methane cannot be prepared from unsaturated hydrocarbons and by Wurtz reaction.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Question 66
Given below are two statements:
Statement I: 3-phenylpropene reacts with HBr and gives secondary alkyl bromide having a chiral carbon atom as the major product.
Statement II: Aryl chlorides and aryl cyanides can be prepared by Sandmeyer reaction as well as Gattermann reaction.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Question 67
Consider the following sequence of reactions
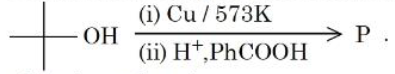
The major product P is:
NTA JEE Mains 6th April Shift 1 2026 - Question 68
Arrange the following compounds according to increasing order of boiling points.
n-C$$_4$$H$$_9$$OH (A), n-C$$_4$$H$$_9$$NH$$_2$$ (B), n-C$$_4$$H$$_{10}$$ (C) and C$$_2$$H$$_5$$NHC$$_2$$H$$_5$$ (D).
NTA JEE Mains 6th April Shift 1 2026 - Question 69
Match the LIST-I with LIST-II
| List-I Deficiency Disease | List-II Vitamin | ||
|---|---|---|---|
| A. | Scurvy | I. | Pyridoxine |
| B. | Convulsions | II. | Vitamin A |
| C. | Cheilosis | III. | Ascorbic Acid |
| D. | Xerophthalmia | IV. | Riboflavin |
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Question 70
Match the LIST-I with LIST-II
| List-I Amino acid | List-II Positive reaction/Test for functional group present in side chain of amino acid | ||
|---|---|---|---|
| A. | Glutamine | I. | Hinsberg's test |
| B. | Lysine | II. | Neutral FeCl$$_3$$ test |
| C. | Tyrosine | III. | Ceric ammonium nitrate test |
| D. | Serine | IV. | Hoffman bromamide degradation |
Choose the correct answer from the options given below:


