NTA JEE Mains 5th April Shift 1 2026
For the following questions answer them individually
NTA JEE Mains 5th April Shift 1 2026 - Question 71
Consider the following species:
$$BrF_5$$, $$XeF_5^-$$, $$BF_4^-$$, $$ICl_4^-$$, $$XeF_4$$, $$SF_4$$, $$NH_4^+$$, $$ClF_3$$, $$XeF_2$$, $$ICl_2^-$$
Number of species having $$sp^3d$$ hybridized central atom is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Question 72
In an estimation of sulphur by Carius method 0.2 g of the substance gave 0.6 g of BaSO$$_4$$. The percentage of sulphur in the substance is _______%. (Given molar mass in g mol$$^{-1}$$ S: 32, BaSO$$_4$$: 231)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Question 73
One mole of phenol is treated with dilute HNO$$_3$$ at 298 K to give a mixture of products. The mixture is separated by steam distillation. The steam volatile compound (X) is separated. The increase in percentage of oxygen in (X) with respect to phenol is _______ $$\times 10^{-1}$$ %. (Given molar mass in g mol$$^{-1}$$ H:1, C:12, N:14, O:16)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Question 74
The values of pressure equilibrium constant recorded at different temperatures for the following equilibrium reaction have been given below
$$A(g) \rightleftharpoons B(g) + C(g)$$
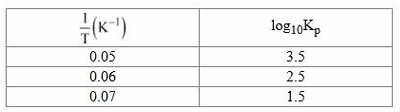
The magnitude of $$\frac{\Delta H^\circ}{R}$$ calculated from the above data is _______. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Question 75
If the half life of a first order reaction is 6.93 minutes then the time required for completion of 99% of the reaction will be _______ minutes. (Given: log 2 = 0.3010)
789
456
123
0.-
Clear All


