NTA JEE Mains 5th April Shift 1 2026
For the following questions answer them individually
NTA JEE Mains 5th April Shift 1 2026 - Question 61
The correct statements about metal carbonyls are
A. The metal-carbon bonds in metal carbonyls possess both $$\sigma$$ and $$\pi$$-character.
B. Due to synergic bonding interactions between metal and CO ligand, the metal-carbon bond becomes weak.
C. The metal-carbon $$\sigma$$ bond is formed by the donation of lone pair of electrons on the carbonyl carbon into a vacant orbital of metal.
D. The metal-carbon $$\pi$$ bond is formed by the donation of electrons from filled d-orbital of metal into vacant $$\pi^*$$ orbital of CO.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Question 62
Given below are two statements:
Statement I: Each electron in $$e_g$$ orbitals destabilizes the orbitals by $$+0.6 \Delta_o$$ and each electron in the $$t_{2g}$$ orbitals stabilizes the orbitals by $$-0.4 \Delta_o$$ in an octahedral field on the basis of crystal field theory.
Statement II: All the d-orbitals of the transition metals have the same energy in their free atomic state but when a complex is formed the ligands destroy the degeneracy of these orbitals on the basis of crystal field theory.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 5th April Shift 1 2026 - Question 63
Given below are two statements:
Statement I: On the basis of inductive effect, the order of stability of alkyl carbanions is $$CH_3^- > CH_3CH_2^- > (CH_3)_2CH^- > (CH_3)_3C^-$$.
Statement II: Allyl and benzyl carbanions are more stabilised by inductive effect and not by resonance effect.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 5th April Shift 1 2026 - Question 64
"P" is a hydrocarbon of molecular formula: $$C_8H_{14}$$. On ozonolysis, "P" forms "Q". "Q" on treatment with alkali under reflux condition produces "R", which on treatment with $$I_2$$/NaOH gives a yellow precipitate. Acidification of the solution gives "S". The structure of "S" is given below:-
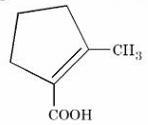
The correct structure of "P" is
NTA JEE Mains 5th April Shift 1 2026 - Question 65
For the following Friedel Craft's alkylation reaction, which of the statements are correct?
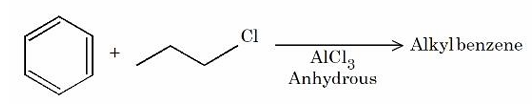
A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Question 66
Benzyl isocyanide can be obtained from:
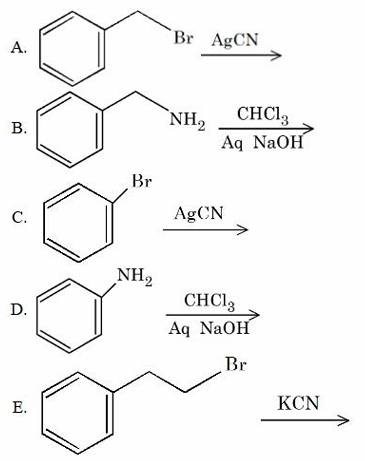
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Question 67
Consider compounds A, B and C with following structural formulae
A = $$CH_3 - CH_2 - CH_2 - CH_2 - CH_2 - OH$$
B = $$CH_2 = CH - CH_2 - CH_2 - CH_3$$
C = $$HO - CH_2 - CH_2 - CH(OH) - CH_3$$
For the conversion of B from A, reagent (D) required is _______ and structural formula of product (E) obtained when C undergoes same reaction using excess reagent (D) is _______.
NTA JEE Mains 5th April Shift 1 2026 - Question 68
Identify the incorrect statements.
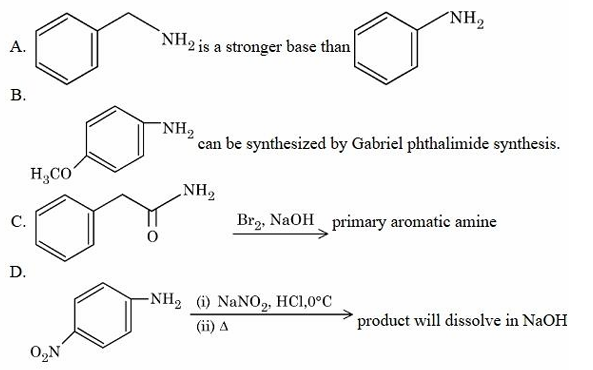
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Question 69
Identify the correct statements.
A. Glucose exists in two anomeric forms.
B. Anomers of glucose differ in configuration at C-1 in cyclic hemiacetal structure.
C. Melting point of $$\alpha$$-anomer of glucose is greater than $$\beta$$-anomer.
D. Specific rotation of $$\alpha$$-anomer is +19° while for $$\beta$$-anomer is +112°.
E. $$\alpha$$ and $$\beta$$-anomers of glucose are prepared by crystallization of saturated glucose solution at 303 K and 371 K respectively.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Question 70
Given below are two statements:
Statement I: Sodium dichromate and potassium dichromate are classified as primary standards in titrimetric analysis.
Statement II: Phenolphthalein is a weak base, therefore it dissociates in acidic medium.
In the light of the above statements, choose the correct answer from the options given below


