NTA JEE Mains 5th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 5th April 2024 Shift 1 - Question 31
An organic compound has $$42.1\%$$ carbon, $$6.4\%$$ hydrogen and remainder is oxygen. If its molecular weight is 342, then its molecular formula is :
NTA JEE Mains 5th April 2024 Shift 1 - Question 32
The incorrect postulates of the Dalton's atomic theory are : (A) Atoms of different elements differ in mass. (B) Matter consists of divisible atoms. (C) Compounds are formed when atoms of different element combine in a fixed ratio. (D) All the atoms of given element have different properties including mass. (E) Chemical reactions involve reorganisation of atoms. Choose the correct answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Question 33
Given below are two statements :
Statement I : In group 13, the stability of +1 oxidation state increases down the group.
Statement II : The atomic size of gallium is greater than that of aluminium.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Question 34
The statement(s) that are correct about the species $$O^{2-}, F^-, Na^+$$ and $$Mg^{2+}$$. (A) All are isoelectronic (B) All have the same nuclear charge (C) $$O^{2-}$$ has the largest ionic radii (D) $$Mg^{2+}$$ has the smallest ionic radii. Choose the most appropriate answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Question 35
Given below are two statements : One is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : Enthalpy of neutralisation of strong monobasic acid with strong monoacidic base is always $$-57 \text{ kJ mol}^{-1}$$. Reason (R) : Enthalpy of neutralisation is the amount of heat liberated when one mole of $$H^+$$ ions furnished by acid combine with one mole of $$OH^-$$ ions furnished by base to form one mole of water. In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Mains 5th April 2024 Shift 1 - Question 36
The following reaction occurs in the Blast furnace where iron ore is reduced to iron metal $$Fe_2O_{3(s)} + 3CO_{(g)} \rightleftharpoons Fe_{(l)} + 3CO_{2(g)}$$. Using the Le-Chatelier's principle, predict which one of the following will not disturb the equilibrium.
NTA JEE Mains 5th April 2024 Shift 1 - Question 37
The number of neutrons present in the more abundant isotope of boron is '$$x$$'. Amorphous boron upon heating with air forms a product, in which the oxidation state of boron is '$$y$$'. The value of $$x + y$$ is ______
NTA JEE Mains 5th April 2024 Shift 1 - Question 38
Number of $$\sigma$$ and $$\pi$$ bonds present in ethylene molecule is respectively :
NTA JEE Mains 5th April 2024 Shift 1 - Question 39
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : Cis form of alkene is found to be more polar than the trans form. Reason (R) : Dipole moment of trans isomer of 2-butene is zero. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Question 40
For the Compounds : (A) $$H_3C-CH_2-O-CH_2-CH_2-CH_3$$
(B) $$H_3C-CH_2-CH_2-CH_2-CH_2-CH_3$$ (
C)
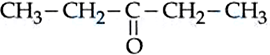
(D)
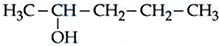
The increasing order of boiling point is :


