NTA JEE Mains 31st Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 41
Identify $$A$$ and $$B$$ in the following reaction sequence:
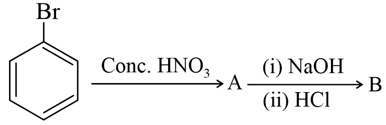
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 42
Choose the correct statements from the following:
A. All group 16 elements form oxides of general formula $$EO_2$$ and $$EO_3$$ where $$E = S, Se, Te$$ and $$Po$$. Both the types of oxides are acidic in nature.
B. $$TeO_2$$ is an oxidising agent while $$SO_2$$ is reducing in nature.
C. The reducing property decreases from $$H_2S$$ to $$H_2Te$$ down the group.
D. The ozone molecule contains five lone pairs of electrons.
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 43
Choose the correct statements from the following:
A. $$Mn_2O_7$$ is an oil at room temperature
B. $$V_2O_4$$ reacts with acid to give $$VO_2^{2+}$$
C. CrO is a basic oxide
D. $$V_2O_5$$ does not react with acid
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 44
Select the option with correct property:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 45
Match List I with List II

Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 46
The correct order of reactivity in electrophilic substitution reaction of the following compounds is:
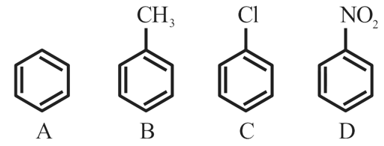
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 47
Identify the name reaction:
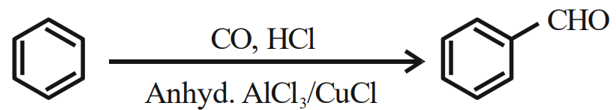
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 48
Identify major product 'P' formed in the following reaction:
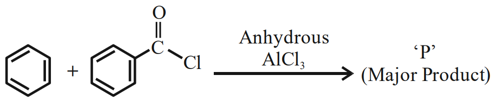
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 49
The azo-dye $$Y$$ formed in the following reactions is: Sulphanilic acid + NaNO$$_2$$ + CH$$_3$$COOH $$\rightarrow$$ X
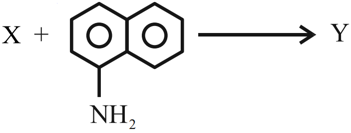
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 50
Given below are two statements:
Statement I: Aniline reacts with con. $$H_2SO_4$$ followed by heating at 453-473 K gives p-aminobenzene sulphonic acid, which gives blood red colour in the 'Lassaigne's test.
Statement II: In Friedel-Craft's alkylation and acylation reactions, aniline forms salt with the $$AlCl_3$$ catalyst. Due to this, nitrogen of aniline acquires a positive charge and acts as deactivating group.
In the light of the above statements, choose the correct answer from the options given below:


