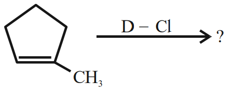NTA JEE Mains 31st Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 31
A sample of CaCO$$_3$$ and MgCO$$_3$$ weighed 2.21 g is ignited to constant weight of 1.152 g. The composition of the mixture is: (Given molar mass in g mol$$^{-1}$$, CaCO$$_3$$: 100, MgCO$$_3$$: 84)
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 32
The four quantum numbers for the electron in the outer most orbital of potassium (atomic no. 19) are
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 33
Consider the following elements.
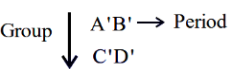
Which of the following is/are true about A', B', C' and D'?
A. Order of atomic radii: $$B' < A' < D' < C'$$
B. Order of metallic character: $$B' < A' < D' < C'$$
C. Size of the element: $$D' < C' < B' < A'$$
D. Order of ionic radii: $$B'^{+} < A'^{+} < D'^{+} < C'^{+}$$
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 34
Which of the following is least ionic?
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 35
$$A_g \rightleftharpoons B_g + \frac{C}{2}_g$$. The correct relationship between $$K_P$$, $$\alpha$$ and equilibrium pressure $$P$$ is
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 36
Given below are two statements:
Statement I: $$S_8$$ solid undergoes disproportionation reaction under alkaline conditions to form $$S^{2-}$$ and $$S_2O_3^{2-}$$
Statement II: $$ClO_4^{-}$$ can undergo disproportionation reaction under acidic condition.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 37
Given below are two statements:
Statement I: Group 13 trivalent halides get easily hydrolysed by water due to their covalent nature.
Statement II: AlCl$$_3$$ upon hydrolysis in acidified aqueous solution forms octahedral $$[Al(H_2O)_6]^{3+}$$ ion.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 38
Identify structure of 2, 3-dibromo-1-phenylpentane.
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 39
The fragrance of flowers is due to the presence of some steam volatile organic compounds called essential oils. These are generally insoluble in water at room temperature but are miscible with water vapour in the vapour phase. A suitable method for the extraction of these oils from the flowers is:
NTA JEE Mains 31st Jan 2024 Shift 2 - Question 40
Major product of the following reaction is: