NTA JEE Mains 30th Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 51
$$0.05 \text{ cm}$$ thick coating of silver is deposited on a plate of area $$0.05 \text{ m}^2$$. The number of silver atoms deposited on plate are _______ $$\times 10^{23}$$. (At mass $$Ag = 108$$, $$d = 7.9 \text{ g cm}^{-3}$$) Round off to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 52
If IUPAC name of an element is "Unununnium" then the element belongs to $$n$$th group of periodic table. The value of $$n$$ is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 53
The total number of molecular orbitals formed from $$2s$$ and $$2p$$ atomic orbitals of a diatomic molecule is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 54
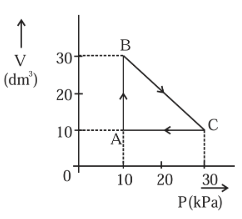
An ideal gas undergoes a cyclic transformation starting from the point $$A$$ and coming back to the same point by tracing the path $$A \rightarrow B \rightarrow C \rightarrow A$$ as shown in the diagram. The total work done in the process is _____ J.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 55
The pH at which $$Mg(OH)_2 [K_{sp} = 1 \times 10^{-11}]$$ begins to precipitate from a solution containing $$0.10 \text{ M } Mg^{2+}$$ ions is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 56
$$2MnO_4^- + bI^- + cH_2O \rightarrow xI_2 + yMnO_2 + zOH^-$$
If the above equation is balanced with integer coefficients, the value of $$z$$ is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 57
On a thin layer chromatographic plate, an organic compound moved by $$3.5 \text{ cm}$$, while the solvent moved by $$5 \text{ cm}$$. The retardation factor of the organic compound is ____________ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 58
The mass of sodium acetate $$(CH_3COONa)$$ required to prepare $$250 \text{ mL}$$ of $$0.35 \text{ M}$$ aqueous solution is _____ g. (Molar mass of $$CH_3COONa$$ is $$82.02 \text{ g mol}^{-1}$$) Round off to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 59
The rate of first order reaction is $$0.04 \text{ mol L}^{-1} \text{s}^{-1}$$ at $$10$$ minutes and $$0.03 \text{ mol L}^{-1} \text{s}^{-1}$$ at $$20$$ minutes after initiation. Half life of the reaction is ______ minutes. (Given $$\log 2 = 0.3010$$, $$\log 3 = 0.4771$$)
Round off your answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 60
The compound formed by the reaction of ethanal with semicarbazide contains _______ number of nitrogen atoms.
789
456
123
0.-
Clear All


