NTA JEE Mains 30th Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 31
Given below are two statements:
Statement-I: The orbitals having same energy are called as degenerate orbitals.
Statement-II: In hydrogen atom, $$3p$$ and $$3d$$ orbitals are not degenerate orbitals.
In the light of the above statements, choose the most appropriate answer from the options given
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 32
Given below are the two statements: one is labeled as Assertion (A) and the other is labeled as Reason (R).
Assertion (A): There is a considerable increase in covalent radius from $$N$$ to $$P$$. However from $$As$$ to $$Bi$$ only a small increase in covalent radius is observed.
Reason (R): covalent and ionic radii in a particular oxidation state increases down the group.
In the light of the above statement, choose the most appropriate answer from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 33
Match List - I with List-II

NTA JEE Mains 30th Jan 2024 Shift 1 - Question 34
Structure of $$4-\text{Methylpent}-2-\text{enal}$$ is:
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 35
Example of vinylic halide is
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 36
Given below are two statements one is labeled as Assertion (A) and the other is labeled as Reason (R).
Assertion (A): $$CH_2=CH-CH_2-Cl$$ is an example of allyl halide
Reason (R): Allyl halides are the compounds in which the halogen atom is attached to $$sp^2$$ hybridised carbon atom.
In the light of the two above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 37
Which of the following molecule/species is most stable?
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 38
Compound $$A$$ formed in the following reaction reacts with $$B$$ gives the product $$C$$. Find out $$A$$ and $$B$$.
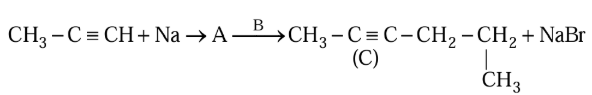
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 39
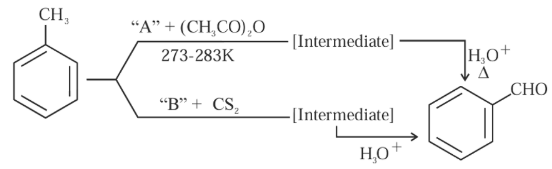
In the given reactions identify the reagent $$A$$ and reagent $$B$$
NTA JEE Mains 30th Jan 2024 Shift 1 - Question 40
What happens to freezing point of benzene when small quantity of naphthalene is added to benzene?


