NTA JEE Mains 29th Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 51

Choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 52
J of energy is transferred as heat to 0.5 mol of Argon gas at 298 K and 1.00 atm. The final temperature and the change in internal energy respectively are: Given : $$R = 8.3 J K^{-1}mol^{-1}$$
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 53
At temperature $$T$$, compound $$AB_{2(g)}$$ dissociates as $$AB_{2} \rightleftharpoons AB_{(g)}+\frac{1}{2}B_{2(g)}$$ having degree of dissociation $$x$$ (small compared to unity). The correct expression for $$x$$ in terms of $$K_{p}$$ and p is
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 54
An element 'E'has the ionisation enthalpy value of $$374 kJ mol^{-1}$$.'E'reacts with elements A, B, C and D with electron gain enthalpy values of −328, −349, −325 and $$-295 kJ mol^{-1}$$, respectively. The correct order of the products EA, EB, EC and ED in terms of ionic character is :
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 55
Total number of nucleophiles from the following is :
$$NH_{3},PhSH,(H_{3}C)_{2}S,H_{2}C=CH_{2},\ominus\\O H,H_{3}O^{\oplus},(CH_{3})_{2}CO,\rightleftharpoons NCH_{3}$$
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 56
The steam volatile compounds among the following are :
(A)
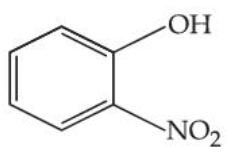
(B)
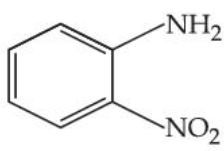
(C)
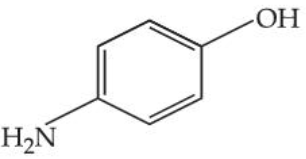
(D)
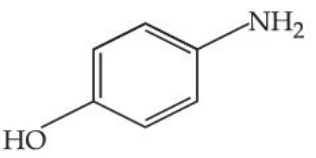
Choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 57
Given below are two statements : Statement (I): The radii of isoelectronic species increases in the order. $$Mg^{2+} < Na^{+} < F^{-} < O^{2-}$$ Statement (II): The magnitude of electron gain enthalpy of halogen decreases in the order. Cl > F > Br > I In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 58
Match List - I with List - II.

choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 59
The molar conductivity of a weak electrolyte when plotted against the square root of its concentration, which of the following is expected to be observed ?
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 60
The standard reduction potential values of some of the p-block ions are given below. Predict the one with the strongest oxidising capacity.


