NTA JEE Mains 29th Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 71
The molar mass of the water insoluble product formed from the fusion of chromite ore $$(FeCr_{2}O_{4})$$ with $$Na_{2}CO_{3}$$ in presence of $$O_{2}$$ is_______$$gmol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 72
Given below are some nitrogen containing compounds
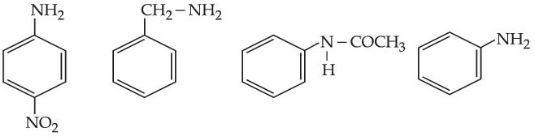
Each of them is treated with HCl separately. 1.0 g of the most basic compound will consume _______ mg of HCl. (Given molar mass in $$gmol^{-1}$$ C : 12, H : 1, O : 16, Cl : 35.5)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 73
The sum of sigma $$(\sigma)$$ and pi$$(\pi)$$ bonds in Hex-1,3-dien-5-yne is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 74
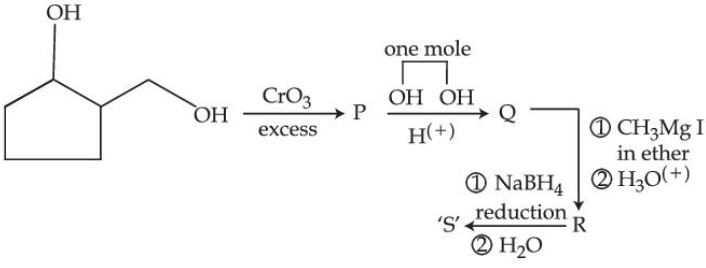
0.1 mole of compound ' S ' will weigh _______ g. (Given molar mass in $$gmol^{-1}$$ C : 12, H : 1, O : 16)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Question 75
If $$A_{2}B$$ is 30% ionised in an aqueous solution, then the value of van't Hoff factor (i) is ______ $$\times 10^{-1}$$
789
456
123
0.-
Clear All


