NTA JEE Mains 29th Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 51
Number of compounds with one lone pair of electrons on central atom amongst following is _______
$$O_3, H_2O, SF_4, ClF_3, NH_3, BrF_5, XeF_4$$
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 52
The number of species from the following which are paramagnetic and with bond order equal to one is _______
$$H_2, He_2^+, O_2^+, N_2^{2-}, O_2^{2-}, F_2, Ne_2^+, B_2$$
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 53
For the reaction $$N_2O_4(g) \rightleftharpoons 2NO_2(g)$$, $$K_p = 0.492$$ atm at $$300$$ K. $$K_c$$ for the reaction at same temperature is _______ $$\times 10^{-2}$$. (Given: $$R = 0.082$$ L atm mol$$^{-1}$$ K$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 54
Number of compounds among the following which contain sulphur as heteroatom is _______.
Furan, Thiophene, Pyridine, Pyrrole, Cysteine, Tyrosine
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 55
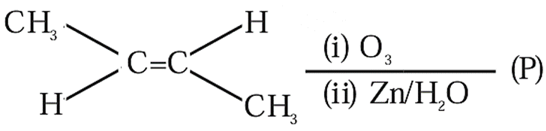
Consider the given reaction. Trans-2-butene $$\xrightarrow{(i) O_3}$$ $$\xrightarrow{(ii) Zn/H_2O}$$ (P)
The total number of oxygen atoms present per molecule of the product (P) is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 56
A solution of $$H_2SO_4$$ is $$31.4\%$$ $$H_2SO_4$$ by mass and has a density of $$1.25$$ g/mL. The molarity of the $$H_2SO_4$$ solution is M (nearest integer) [Given molar mass of $$H_2SO_4 = 98$$ g mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 57
The osmotic pressure of a dilute solution is $$7 \times 10^5$$ Pa at $$273$$ K. Osmotic pressure of the same solution at $$283$$ K is _______ $$\times 10^4$$ Nm$$^{-2}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 58
The mass of zinc produced by the electrolysis of zinc sulphate solution with a steady current of $$0.015$$ A for $$15$$ minutes is _______ $$\times 10^{-4}$$ g. (Atomic mass of zinc $$= 65.4$$ amu)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 59
For a reaction taking place in three steps at same temperature, overall rate constant $$K = \frac{K_1 K_2}{K_3}$$. If $$Ea_1, Ea_2$$ and $$Ea_3$$ are $$40, 50$$ and $$60$$ kJ/mol respectively, the overall $$Ea$$ is _______ kJ/mol.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 60
From the compounds given below, number of compounds which give positive Fehling's test is _______
Benzaldehyde, Acetaldehyde, Acetone, Acetophenone, Methanal, 4-nitrobenzaldehyde, cyclohexane carbaldehyde.
789
456
123
0.-
Clear All


