NTA JEE Mains 29th Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 31
The correct set of four quantum numbers for the valence electron of rubidium atom $$(Z = 37)$$ is:
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 32
Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: The first ionisation enthalpy decreases across a period.
Reason R: The increasing nuclear charge outweighs the shielding across the period.
In the light of the above statements, choose the most appropriate from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 33
Which of the following is not correct?
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 34
Chlorine undergoes disproportionation in alkaline medium as shown below :
$$aCl_2(g) + bOH^-(aq) \rightarrow cClO^-(aq) + dCl^-(aq) + eH_2O(l)$$
The values of $$a, b, c$$ and $$d$$ in a balanced redox reaction are respectively :
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 35
$$KMnO_4$$ decomposes on heating at $$513$$ K to form $$O_2$$ along with
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 36
Given below are two statements :
Statement I : The electronegativity of group 14 elements from Si to Pb gradually decreases.
Statement II : Group 14 contains non-metallic, metallic, as well as metalloid elements.
In the light of the above statements, choose the most appropriate from the options given below :
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 37
The interaction between $$\pi$$ bond and lone pair of electrons present on an adjacent atom is responsible for
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 38
The difference in energy between the actual structure and the lowest energy resonance structure for the given compound is:
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 39
Appearance of blood red colour, on treatment of the sodium fusion extract of an organic compound with $$FeSO_4$$ in presence of concentrated $$H_2SO_4$$ indicates the presence of element/s
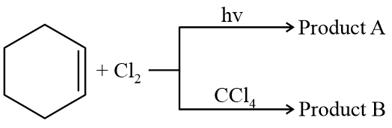
NTA JEE Mains 29th Jan 2024 Shift 1 - Question 40
Identify product A and product B