NTA JEE Mains 28th Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 61
Method used for separation of mixture of products (B and C) obtained in the following reaction is
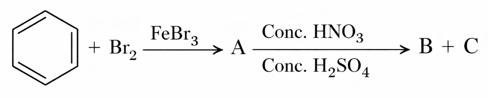
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 62
Given below are two statements for the following reaction sequence.
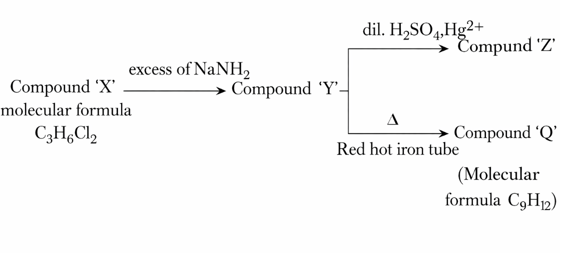
Statement I: Compound 'Z' will give yellow precipitate with NaOI.
Statement II: Compound 'Q' has two different types of'H' atoms (aromatic:aliphatic) in the ratio 1 :3.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 63
At T(K), 2 moles of liquid A and 3 moles of liquid B are mixed. The vapour pressure of ideal solution fonned is 320 mm Hg. At this stage, one mole of A and one mole of B are added to the solution. The vapour pressure is now measmed as 328.6 mm Hg. The vapom pressure (in mm Hg) of A and B are respectively:
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 64
Given below are the four isomeric compounds (P, Q, R, S)
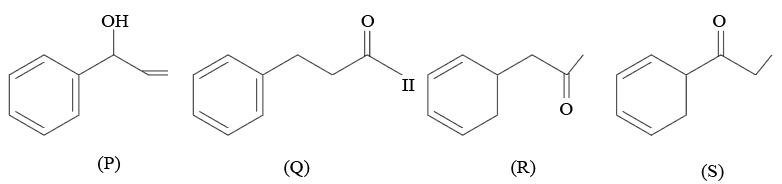
Identify correct statements from below
A. Q, R and S will give precipitate with 2, 4 - DNP.
B. P and Q will give positive Bayer's test.
C. Q and R will give sooty flame.
D. Rand S will give yellow precipitate with $$I_{2}/NaOH$$.
E. Q alone will deposit silver with Tollen's reagent.
Choose the correct option.
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 65
The wave numbers of three spectral lines of H atom are considered. Identify the set of spectral lines belonging to Balmer series.
(R = Rydberg constant)
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 66
Given below are two statements:
Statement I: The number of species among $$BF_{4}^{-},SiF_{4},XeF_{4}\text{ and }SF_{4}$$,that have unequal E-F bond lengths is two. Here, E is the central atom.
Satement II: Among $$O_{2}^{-},O_{2}^{2-},F_{2}\text{ and }O_{2}^{+},O_{2}^{-}$$ has the highest bond order.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 67
CORRECT order of stability for the following is
$$CH_{2}=CH^{-},CH_{3}-CH_{2}^{-},CH\equiv C^{-}$$
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 68
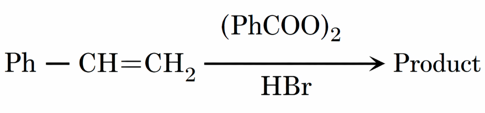
Consider the above reaction
A. The reaction proceeds through a more stable radical intermediate.
B. The role of peroxide is to generate H^{.} (Hydrogen radical).
C. During this reaction, benzene is formed as a byproduct.
D. 1-Bromo-2- phenylethane is formed as the minor product.
E. The same reaction in absence of peroxide proceeds via carbocation intermediate.
Identify the correct statements. Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 69
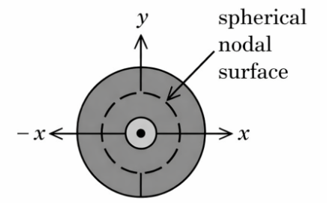
Figure 1. electron probability density for 2s orbital
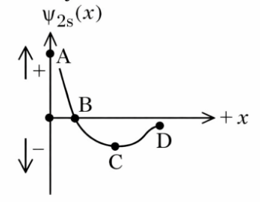
Figure 2. wave function for 2s orbital
Which of the following point in Figure 2 most accurately represents the nodal surface as shown in Figure 1?
NTA JEE Mains 28th Jan 2026 Shift 1 - Question 70
Regarding the hydrides of group 15 elements $$EH_{3}$$(E = N, P, As, Sb), select the correct statement from the following:
A. The stability of hydrides decreases down the group.
B. The basicity of hydrides decreases down the group.
C. The reducing character increases down the group.
D. The boiling point increases down the group.
Choose the correct answer from the options given below:


