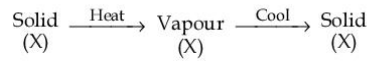NTA JEE Mains 28th Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 51
Concentrated nitric acid is labelled as 75% by mass. The volume in mL of the solution which contains 30 g of nitric acid is ____. Given : Density of nitric acid solution is 1.25 g/mL .
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 52
Given below are two statements : Statement (I) :
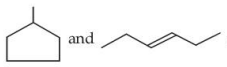
are isomeric compounds. Statement (II) :
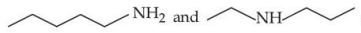
are functional group isomers.
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 53
Identify product [A],[B] and [C] in the following reaction sequence.
$$\mathrm{CH_3 - C \equiv CH \xrightarrow[\mathrm{H_2}]{\mathrm{Pd/C}} [A] \xrightarrow[\mathrm{(ii)\ Zn,\ H_2O}]{\mathrm{(i)\ O_3}} [B] + [C]}$$
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 54
The total number of compounds from below when treated with hot $$KMnO_{4}$$ giving benzoic acid is :
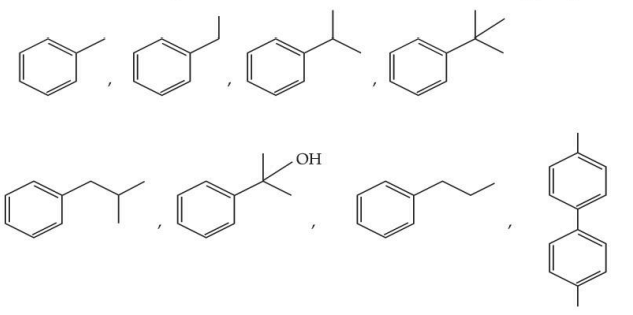
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 55
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 56
Identify correct statements : (A) Primary amines do not give diazonium salts when treated with $$NaNO_{2}$$ in acidic condition. (B) Aliphatic and aromatic primary amines on heating with $$CHCl_{3}$$and ethanolic KOH form carbylamines. (C) Secondary and tertiary amines also give carbylamine test. (D) Benzenesulfonyl chloride is known as Hinsberg's reagent. (E) Tertiary amines reacts with benzenesulfonyl chloride very easily. Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 57
For bacterial growth in a cell culture, growth law is very similar to the law of radioactive decay. Which of the following graphs is most suitable to represent bacterial colony growth ? Where N - Number of Bacteria at any time, $$N_{0}$$ - Initial number of Bacteria.
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 58
The amphoteric oxide among $$V_{2}O_{3}$$ and $$V_{2}O_{5}$$ , upon reaction with alkali leads to formation of an oxide anion. The oxidation state of V in the oxide anion is :
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 59
Arrange the following in increasing order of solubility product :$$\mathrm{Ca(OH)_2, \ AgBr, \ PbS, \ HgS}$$
NTA JEE Mains 28th Jan 2025 Shift 2 - Question 60
The purification method based on the following physical transformation is :