NTA JEE Mains 27th Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 51
Volume of 3M NaOH (formula weight 40 g mol$$^{-1}$$) which can be prepared from 84 g of NaOH is _____ $$\times 10^{-1}$$ dm$$^3$$.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 52
9.3 g of aniline is subjected to reaction with excess of acetic anhydride to prepare acetanilide. The mass of acetanilide produced if the reaction is 100% completed is _____ $$\times 10^{-1}$$ g.
(Given molar mass in g mol$$^{-1}$$: N = 14, O = 16, C = 12, H = 1)
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 53
1 mole of PbS is oxidised by X moles of $$O_3$$ to get Y moles of $$O_2$$. X + Y =
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 54
Total number of ions from the following with noble gas configuration is
$$Sr^{2+}$$ (Z = 38), $$Cs^+$$ (Z = 55), $$La^{2+}$$ (Z = 57), $$Pb^{2+}$$ (Z = 82), $$Yb^{2+}$$ (Z = 70) and $$Fe^{2+}$$ (Z = 26)
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 55
The number of non-polar molecules from the following is
$$HF$$, $$H_2O$$, $$SO_2$$, $$H_2$$, $$CO_2$$, $$CH_4$$, $$NH_3$$, $$HCl$$, $$CHCl_3$$, $$BF_3$$
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 56
For a certain thermochemical reaction $$M \rightarrow N$$ at $$T = 400$$ K, $$\Delta H^o = 77.2$$ kJ mol$$^{-1}$$, $$\Delta S^o = 122$$ J K$$^{-1}$$, log equilibrium constant ($$\log K$$) is $$-$$ _____ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 57
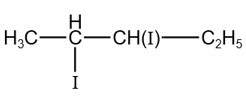
Total number of compounds with Chiral carbon atoms from following is
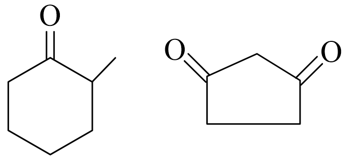
$$CH_3-CH_2-CHNO_2-COOH$$
$$CH_3-CH(I)-CH_2-NO_2$$
$$CH_3-CH_2-CHBr-CH_2-CH_3$$
$$CH_3-CH_2-CH(OH)-CH_2OH$$
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 58
The hydrogen electrode is dipped in a solution of pH = 3 at 25°C. The potential of the electrode will be $$-$$ _____ $$\times 10^{-2}$$ V.
$$\frac{2.303RT}{F} = 0.059$$ V. Round off the answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 59
Time required for completion of 99.9% of first order reaction is _____ times of half life ($$t_{1/2}$$) of the reaction
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 60
The Spin only magnetic moment value of square planar complex $$[Pt(NH_3)_2Cl(NH_2CH_3)]Cl$$ is _____ B.M.
(Nearest integer)
(Given atomic number for Pt = 78)
789
456
123
0.-
Clear All


