NTA JEE Mains 27th Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 41
Choose the correct option having all the elements with $$d^{10}$$ electronic configuration from the following:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 42
Given below are two statements:
Statement (I) : In the Lanthanoids, the formation of $$Ce^{+4}$$ is favoured by its noble gas configuration.
Statement (II) : $$Ce^{+4}$$ is a strong oxidant reverting to the common +3 state.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 43
Identity the incorrect pair from the following:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 44
Identify from the following species in which $$d^2sp^3$$ hybridization is shown by central atom:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 45
Which among the following halide/s will not show $$S_N1$$ reaction:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 46
Identify B formed in the reaction.
$$Cl-(CH_2)_4-Cl \xrightarrow{excess\ NH_3} A \xrightarrow{NaOH} B + H_2O + NaCl$$
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 47
Phenolic group can be identified by a positive:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 48
Match List-I with List-II.
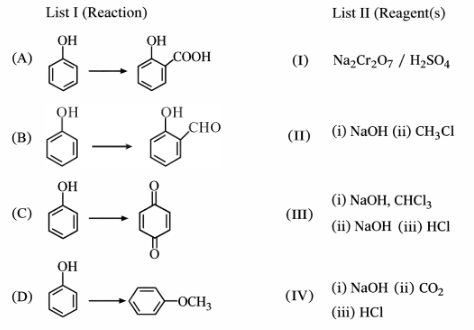
Choose the correct answer from the options given below:
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 49
Major product formed in the following reaction is a mixture of:
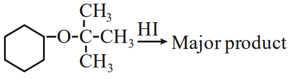
NTA JEE Mains 27th Jan 2024 Shift 2 - Question 50
Which structure of protein remains intact after coagulation of egg white on boiling?


