NTA JEE Mains 24th Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 61
{The conditions and consequence that favours the $$t_{2gg} e_g^1$$ configuration in a metal complex are:
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 62
In the given structure, the number of $$sp \text{ and } sp^2 $$ hybridized carbon atoms present respectively are:
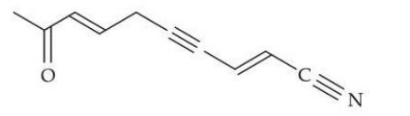
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 63
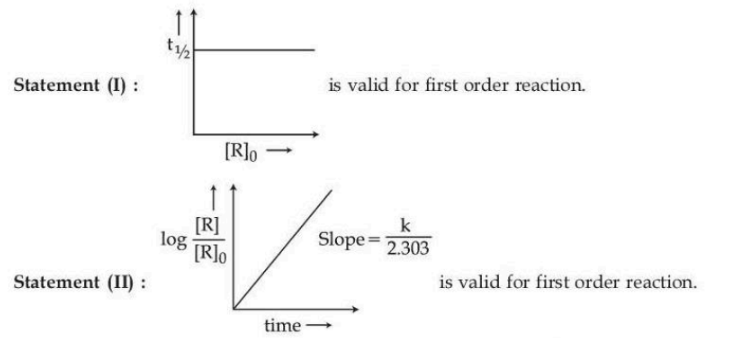
Given below are two statements :
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 64
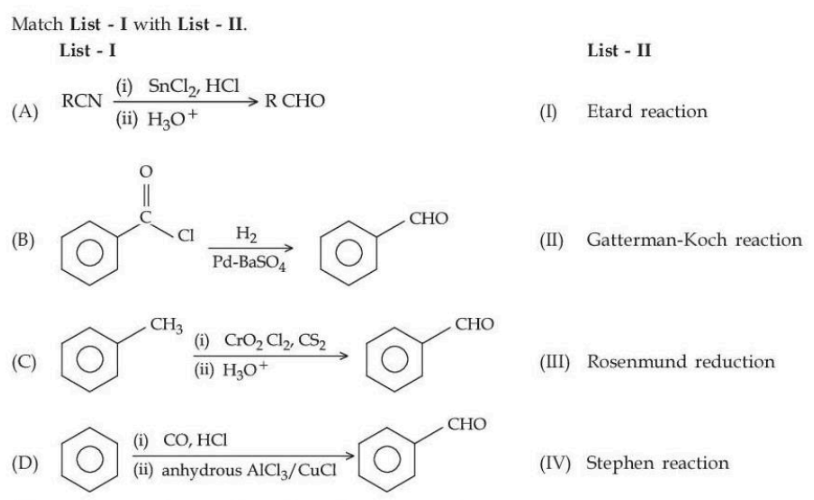
Choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 65
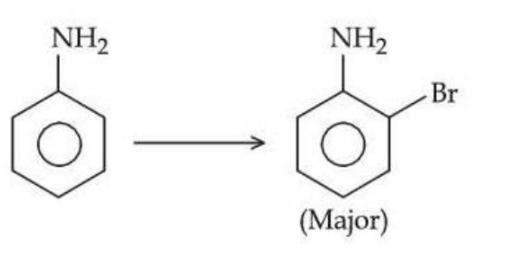
The structure of the major product formed in the following reaction is :
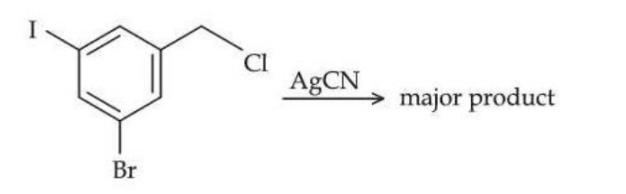
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 66
For reaction
The correct order of set of reagents for the above conversion is :
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 67
The successive 5 ionisation energies of an element are 800, 2427, 3658, 25024 and 32824 kJ/mol, respectively. By using the above values predict the group in which the above element is present:
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 68
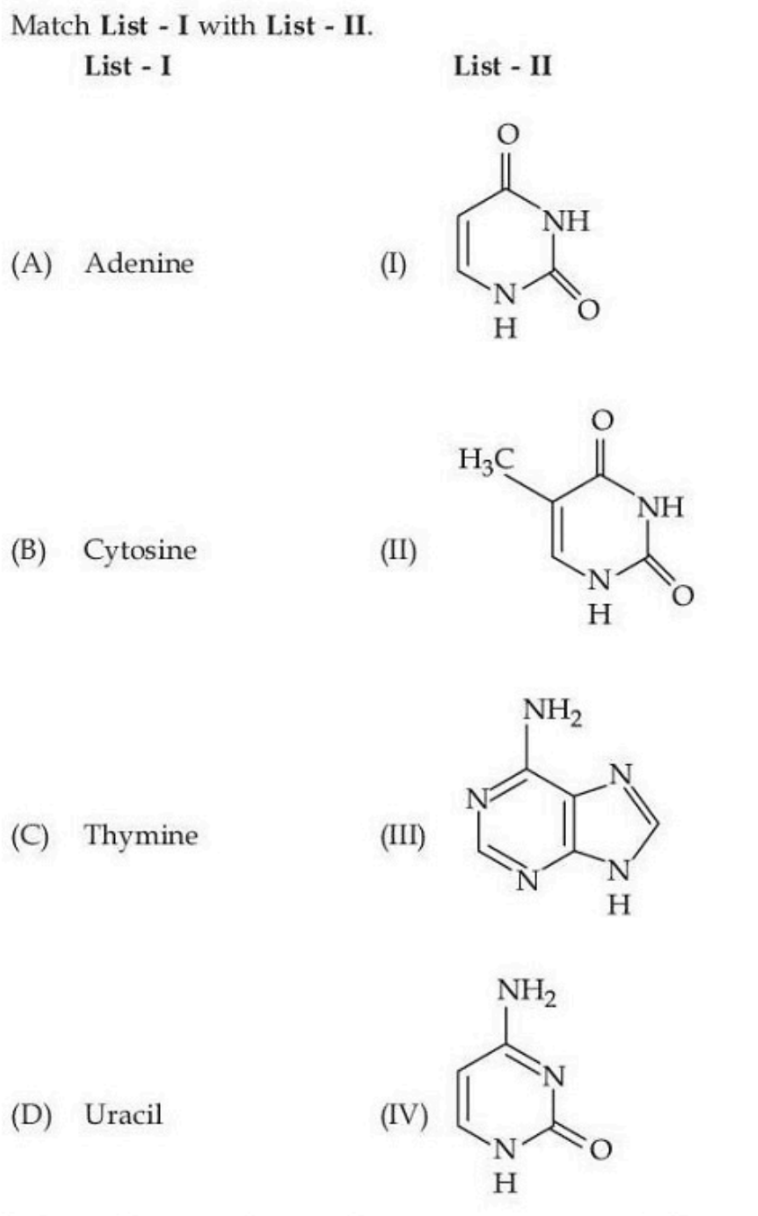
Choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 69
Which of the following mixing of 1 M base and 1 M acid leads to the largest increase in temperature?
NTA JEE Mains 24th Jan 2025 Shift 2 - Question 70
$$S(g) + \frac{3}{2}O_2(g) \rightarrow SO_3(g) + 2x\,\text{kcal} \\SO_2(g) + \frac{1}{2}O_2(g) \rightarrow SO_3(g) + y\,\text{kcal}\\\text{The heat of formation of } SO_2(g) \text{ is given by:}$$


