NTA JEE Mains 24th Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 61
$$ K_{sp} \text{ for } Cr(OH)_3 \text{ is } 1.6\times10^{-30}.$$ What is the molar solubility of this salt in water?
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 62
Which of the following statements are NOT true about the periodic table? A. The properties of elements are function of atomic weights. B. The properties of elements are function of atomic numbers. C. Elements having similar outer electronic configurations are arranged in same period. D. An element's location reflects the quantum numbers of the last filled orbital. E. The number of elements in a period is same as the number of atomic orbitals available in energy level that is being filled. Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 63
Given below are two statements I and II. Statement I: Dumas method is used for estimation of "Nitrogen" in an organic compound. Statement II: Dumas method involves the formation of ammonium sulphate by heating the organic compound with conc $$H_2 SO_4 $$. In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 64
Which of the following statement is true with respect to $$H_2O,\ NH_3$$ and $$CH_4?$$ $$A.$$ The central atoms of all the molecules are $$sp^3$$ hybridized. $$B.$$ The $$H-O-H,\ H-N-H$$ and $$H-C-H$$ angles in the above molecules are $$104.5^\circ,\ 107.5^\circ$$ and $$109.5^\circ$$ respectively. $$C.$$ The increasing order of dipole moment is $$CH_4 < NH_3 < H_2O.$$ $$D.$$ Both $$H_2O$$ and $$NH_3$$ are Lewis acids and $$CH_4$$ is a Lewis base. $$E.$$ A solution of $$NH_3$$ in $$H_2O$$ is basic. In this solution $$NH_3$$ and $$H_2O$$ act as Lowry-Bronsted acid and base respectively. Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 65
Which one of the carbocations from the following is most stable?
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 66
Following are the four molecules "P", "Q", "R" and "S". Which one among the four molecules will react with
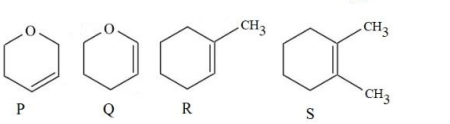
$$H-Br_{(aq)}$$ at the fastest rate?
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 67
For the given cell $$Fe^{2+}_{(aq)} + Ag^+_{(aq)} \rightarrow Fe^{3+}_{(aq)} + Ag_{(s)},$$ the standard cell potential of the above reaction is Given: $$\begin{aligned}Ag^+ + e^- &\rightarrow Ag \qquad E^\circ = x\,V \\Fe^{2+} + 2e^- &\rightarrow Fe \qquad E^\circ = y\,V \\Fe^{3+} + 3e^- &\rightarrow Fe \qquad E^\circ = z\,V \end{aligned}$$
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 68
The large difference between the melting and boiling points of oxygen and sulphur may be explained on the basis of
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 69
Consider the given plots of vapour pressure (VP) vs temperature (T/K). Which amongst the following options is correct graphical representation showing $$\Delta T_f,$$ depression in the freezing point of a solvent in a solution?
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 70
Which of the following linear combination of atomic orbitals will lead to formation of molecular orbitals in homonuclear diatomic molecules [internuclear axis in $$z$$-direction] ? A. $$2p_z$$ and $$2p_x$$ B. 2 s and $$2p_x$$ C. 3 $$d_{xy}$$ and 3 $$d_{x^{2} - y^{2}}$$ D. 2 s and $$2p_z$$ E. $$2p_z$$ and $$3d_{x}^{2}- y^{2}$$ Choose the correct answer from the options given below:


