NTA JEE Mains 24th Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 51
The carbohydrate "Ribose" present in DNA, is A. A pentose sugar B. present in pyranose from C. in "D" configuration D. a reducing sugar, when free E. in $$\alpha$$-anomeric form Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 52
Given below are two statements: Statement I: The conversion proceeds well in the less polar medium.
$$\mathrm{CH_3-CH_2-CH_2-CH_2-Cl} \;\xrightarrow{\;\;HO^-\;\;} \mathrm{CH_3-CH_2-CH_2-CH_2-OH} + \mathrm{Cl^{(-)}}$$
Statement II: The conversion proceeds well in the more polar medium.
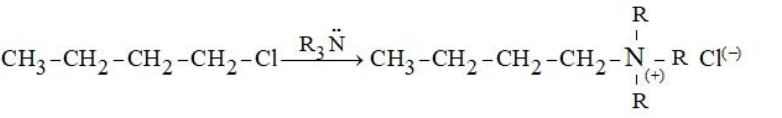
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 53
The product (A) formed in the following reaction sequence is
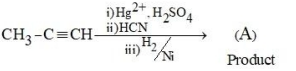
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 54
Aman has been asked to synthesise the molecule
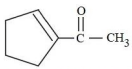
(x).He thought of preparing the molecule using an aldol condensation reaction. He found a few cyclic alkenes in his laboratory. He thought of performing ozonolysis reaction on alkene to produce a dicarbonyl compound followed by aldol reaction to prepare " x ". Predict the suitable alkene that can lead to the formation of " x ".
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 55
Which of the following arrangements with respect to their reactivity in nucleophilic addition reaction is correct?
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 56
Let us consider an endothermic reaction which is non-spontaneous at the freezing point of water. However, the reaction is spontaneous at boiling point of water. Choose the correct option.
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 57
Preparation of potassium permanganate from $$MnO_2$$ involves two step process in which the 1st step is a reaction with $$KOH$$ and $$KNO_3$$ to produce:
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 58
For a reaction, $$N_2O_5(g) \rightarrow 2NO_2(g) + \frac{1}{2}O_2(g)$$ in a constant volume container, no products were present initially. The final pressure of the system when $$50\%$$ of reaction gets completed is:
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 59
One mole of the octahedral complex compound $$Co(NH_3)_5Cl_3$$ gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with excess $$AgNO_3$$ solution to yield two moles of $$AgCl(s).$$ The structure of the complex is:
NTA JEE Mains 24th Jan 2025 Shift 1 - Question 60
Which of the following ions is the strongest oxidizing agent? (Atomic Number of $$Ce = 58,\; Eu = 63,\; Tb = 65,\; Lu = 71$$)


