NTA JEE Mains 24th january 2023 Shift 1
For the following questions answer them individually
NTA JEE Mains 24th january 2023 Shift 1 - Question 51
If wavelength of the first line of the Paschen series of hydrogen atom is 720 nm, then the wavelength of the second line of this series is _______ nm. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 52
For independent process at 300 K.

The number of non-spontaneous process from the following is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 53
The dissociation constant of acetic acid is $$x \times 10^{-5}$$. When 25 mL of 0.2 M $$CH_3COONa$$ solution is mixed with 25 mL of 0.02 M $$CH_3COOH$$ solution, the pH of the resultant solution is found to be equal to 5. The value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 54
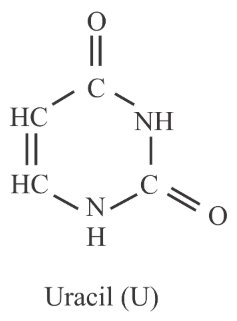
Uracil is base present in RNA with the following structure. % of N in uracil is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 55
When $$Fe_{0.93}O$$ is heated in presence of oxygen, it converts to $$Fe_2O_3$$. The number of correct statement/s from the following is ______.
A. The equivalent weight of $$Fe_{0.93}O$$ is $$\frac{\text{Molecular weight}}{0.79}$$
B. The number of moles of $$Fe^{2+}$$ and $$Fe^{3+}$$ in 1 mole of $$Fe_{0.93}O$$ is 0.79 and 0.14 respectively.
C. $$Fe_{0.93}O$$ is metal deficient with lattice comprising of cubic closed packed arrangement of $$O^{2-}$$ ions.
D. The % composition of $$Fe^{2+}$$ and $$Fe^{3+}$$ in $$Fe_{0.93}O$$ is 85% and 15% respectively.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 56
5 g of NaOH was dissolved in deionized water to prepare a 450 mL stock solution. What volume (in mL) of this solution would be required to prepare 500 mL of 0.1 M solution? Given : Molar Mass of Na, O and H is 23, 16 and 1 g mol$$^{-1}$$ respectively
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 57
At 298 K, a 1 litre solution containing 10 mmol of $$Cr_2O_7^{2-}$$ and 100 mmol of $$Cr^{3+}$$ shows a pH of 3.0. Given : $$Cr_2O_7^{2-} \to Cr^{3+}$$; $$E^0 = 1.330$$ V and $$\frac{2.303RT}{F} = 0.059$$ V. The potential for the half cell reaction is $$x \times 10^{-3}$$ V. The value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 58
The number of correct statement/s from the following is _____.
A. Larger the activation energy, smaller is the value of the rate constant.
B. The higher is the activation energy, higher is the value of the temperature coefficient.
C. At lower temperatures, increase in temperature causes more change in the value of k than at higher temperature.
D. A plot of $$\ln k$$ vs $$\frac{1}{T}$$ is a straight line with slope equal to $$-\frac{E_a}{R}$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 59
The d-electronic configuration of $$CoCl_4^{2-}$$ in tetrahedral crystal field is $$e^m t_2^n$$. The sum of m and number of unpaired electrons is
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Question 60
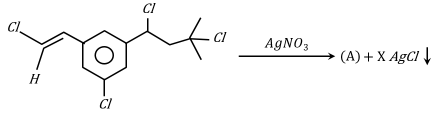
Number of moles of AgCl formed in the following reaction is _____.
789
456
123
0.-
Clear All


