NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 51
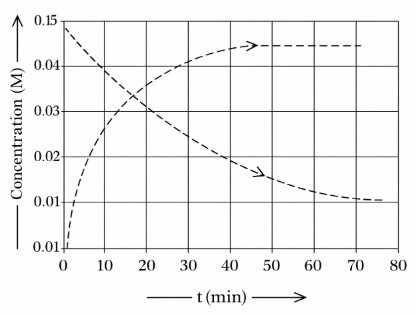
Given above is the concentration vs time plot for a dissociation reaction : $$A \rightarrow nB$$ .
Based on the data of the initial phase of the reaction (initial 10 min), the value of n is________.
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 52
It is noticed that $$Pb^{2+}$$ is more stable than $$Pb^{4+}$$ but $$Sn^{2+}$$ is less stable than $$Sn^{4+}$$
Observe the following reactions.
$$ PbO_2+Pb\rightarrow 2PbO;\triangle_rG^{o}(1) $$
$$ SnO_2+Sn\rightarrow 2SnO;\triangle_rG^{o}(2) $$
Identify the correct set from the following
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 53
Elements X and Y belong to Group 15 . The difference between the electronegativity values of 'X' and phosphorus is higher than that of the difference between phosphorus and 'Y'. 'X' & 'Y' are respectively
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 54
Given below are two statements:
Statement I: The second ionisation enthalpy of Na is larger than the corresponding ionisation enthalpy of Mg.
Statement II: The ionic radius of $$O^{2-}$$ is larger than that of $$F^{-}$$.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 55
Both human DNA and RNA are chiral molecules. The chirality in DNA and RNA arises due to the presence of
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 56
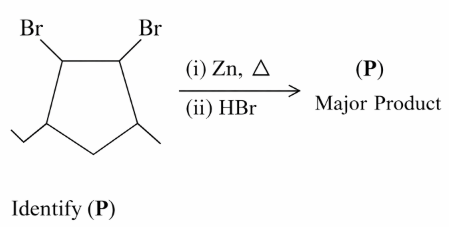
A mixed ether (P), when heated with excess of hot concentrated hydrogen iodide produces two different alkyl iodides which when treated with aq. NaOH give compounds (Q) and (R). Both (Q) and (R) give yellow precipitate with NaOI. Identify the mixed ether (P):
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 57
Identify the CORRECT set of details from the following:
A. $$[Co(NH_3)_6]^{3+}$$ : Inner orbital compex; $$d^{2}sp^{3}$$ hybridized
B. $$[MnCl_6]^{3-}$$ : Outer orbital complex; $$sp^{3}d^{2}$$ hybridized
C. $$[CoF_6]^{3-}$$ : Outer orbital complex; $$d^{2}sp^{3}$$ hybridized
D. $$[FeF_6]^{3-}$$ : Outer orbital complex; $$sp^{3}d^{2}$$ hybridized
E. $$[Ni(CN)_4]^{2-}$$ : Inner orbital complex; $$sp^{3}d^{2}$$ hybridized
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 58
A student has been given a compound "x" of molecular formula- $$C_{6}H_{7}N$$. 'x' is sparingly soluble in water. However, on addition of dilute mineral acid, 'x' becomes soluble in water. 'x' when treated with $$CHCl_{3}$$ and KOH(alc), 'Y' is produced. 'y' has a specific unpleasant smell. On treatment with benzenesulphonyl chloride, 'x' gives a compound 'z' which is soluble in alkali. The number of different "H" atoms present in 'z' is :-
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 59
Iodoform test can differentiate between
A. Methanol and Ethanol
B. $$CH_{3}COOH$$ and $$CH_{3}CH_{2}COOH$$
C. Cyclohexene and cyclohexanone
D. Diethyl ether and Pentan-3-one
E. Anisole and acetone
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 60
The work functions of two metals ($$M_{A}$$ and $$M_{B}$$ ) are in the 1 : 2 ratio. When these metals are exposed to photons of energy 6 eV, the kinetic energy of liberated electrons of $$M_{A}$$ : $$M_{B}$$ is in the ratio of 2.642 : 1. The work functions (in eV) of $$M_{A}$$ and $$M_{A}$$ are respectively.
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 61
Identify the INCORRECT statements from the following:
A. Notation $$_{12}^{24}Mg$$ represents 24 protons and 12 neutrons.
B. Wavelength of a radiation of frequency $$ 4.5\times10^{15}s^{-1} $$ is $$ 6.7\times10^{-8} $$m.
C. One radiation has wavelength =$$\lambda_1(900nm) $$ and energy= $$E_{1}$$. Other radiation has wavelength = $$\lambda_2(300nm) $$ and energy= $$E2 \cdot E1 : E_2= 3 : 1$$.
D. Number of photons of light of wavelength 2000 pm that provides 1 J of
energy is $$1.006 x 10^{16}$$.
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 62
Given below are two statements:
Statement I: $$(CH_3)_3C^+$$ is more stable than $$CH_3^+$$ as nine hyperconjugation interactions are possible in $$(CH_3)_3C^+$$.
Statement II: $$CH_3^+$$ is less stable than $$(CH_3)_3C^+$$ as only three hyperconjugation interactions are possible in $$CH_3^+$$.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 64
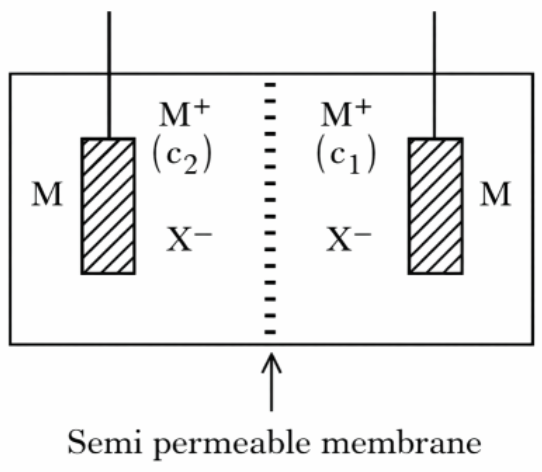
Consider the above electrochemical cell where a metal electrode (M) is undergoing redox reaction by forming $$M^{+}(M\rightarrow M^{+}+e^{-})$$. The cation $$M^{+}$$ is present in two different concentrations $$c_{1}$$ and $$c_{2}$$ as shown above. which of the following statement is correct for generating a positive cell potential?
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 65
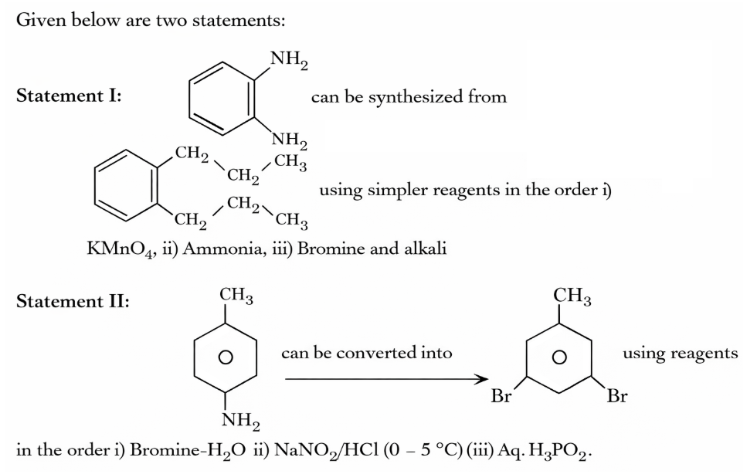
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 66
In Carius method 0.2425 g of an organic compound gave 0.5253 g silver chloride.
The percentage of chlorine in the organic compound is
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 67
Which statements are NOT TRUE about $$XeO_2 F_2$$?
A. It has a see-saw shape.
B. Xe has 5 electron pairs in its valence sheU in XeO 2 F 2.
C. The $$O - Xe- O$$ bond angle is close to $$180^{o}$$.
D. The $$F- Xe -F$$ bond angle is close to $$180^{o}$$.
E. $$Xe$$ has 16 valence electrons in $$XeO_2 F_2$$.
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 68
Observe the following reactions at T(K).
I. $$A\rightarrow$$products
II. $$5Br^{-}(aq)+BrO_{3}\text{ } ^{-}(aq)\rightarrow 3Br_2(aq)+3H_2O(l)$$
Both the reactions are started at 10.00 am. The rates of these reactions at 10.10 am are same. The value of $$-\frac{\triangle[Br^{-}]}{\triangle t}$$ at 10.10am. is $$2\times 10^{-4} mol \text{ }L^{-1}min^{-1}$$. The concentration of A at 10.10am is $$10^{-1}mol \text{ } L^{-1}$$. What is the first order rate constant (in min^{-1}) of reaction I?
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 69
Which of the following statements are TRUE about Haloform reaction?:
A. Sodium hypochlorite reacts with KI to give KOI.
B. KOI is a reducing agent.
C. $$ \alpha,\beta$$-unsaturated methylketone $$\mathrm{CH_3 - CH = CH - C(=O) - CH_3}$$ will give iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 70
The oxidation state of chromium in the final product formed in the reaction between $$KI$$ and acidified $$K_2 Cr_2 O_7$$ solution is:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 71
$$X_2(g ) + Y_2(g ) \rightleftharpoons 2Z(g)$$
$$X_2(g )$$ and $$Y_2(g )$$ are added to a 1 L flask and it is found that the system attains the above equilibrium at T(K) with the number of moles of $$X_2(g ),\text{ } Y_2(g )$$ and $$Z(g)$$ being 3, 3 and 9 mol respectively (equilibrium moles). Under this condition of equilibrium, 10 mol of $$Z(g$$) is added to the flask and the temperature is maintained at $$T(K)$$. Then the number of moles of $$Z(g)$$ in the flask when the new equilibrium is established is __ . (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 72
Consider the following reaction of benzene.
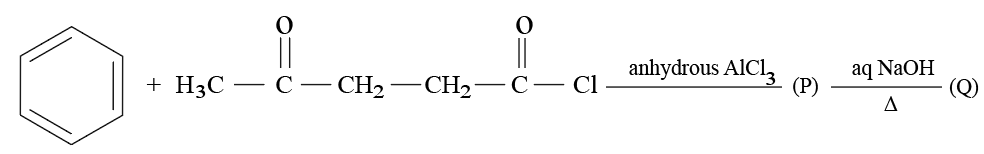
In compound (Q), the percentage of oxygen is ___ %. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 73
200 cc of $$ x\times 10^{-3} M$$ potassium dichromate is required to oxidise 750 cc of 0.6 M Mohr's salt solution in acidic mediun.
Here $$x$$ =___________
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 74
Two liquids A and B form an ideal solution. At 320 K, the vapour pressure of the solution, containing 3 mol of A and 1 mol of B is 500 mm Hg. At the same
temperature, if 1 mol of A is farther added to this solution, vapour pressure of the solution increases by 20 mm Hg. Vapour pressure (in mm Hg) of B in pure state is ____ . (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Chemistry - Question 75
Total number of unpaired electrons present in the central met al atoms/ions of
$$[Ni(CO)_4],[NiCl_4]^{2-},[PtCl_2(NH_3)_2],[Ni(CN)_4]^{2-}$$ and $$[Pt(CN)_4]^{2-}$$ is_____
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)