NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 51
Radius of the first excited state of Helium ion is given as : $$a_0\rightarrow$$ radius of first stationary state of hydrogen atom.
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 52
The incorrect statements regarding geometrical isomerism are : (A) Propene shows geometrical isomerism. (B) Trans isomer has identical atoms/groups on the opposite sides of the double bond. (C) Cis-but-2-ene has higher dipole moment than trans-but-2-ene. (D) 2-methylbut-2-ene shows two geometrical isomers. (E) Trans-isomer has lower melting point than cis isomer. Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 53
A liquid when kept inside a thermally insulated closed vessel $$25^{o}C$$ at was mechanically stirred from outside. What will be the correct option for the following thermodynamic parameters ?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 54
Which of the following electronegativity order is incorrect?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 55
Lanthanoid ions with $$4f^{7} configuration are : $$(A)Eu^{2+}$$\text{ }$$ (B)Gd^{3+}$$\text{ }$$ (C)Eu^{3+}$$\text{ }$$ (D)Tb^{3+}$$\text{ }$$ (E)Sm^{3+}$$ Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 56
Given below are two statements : Statement I : One mole of propyne reacts with excess of sodium to liberate half a mole of $$H_2 $$ gas. Statement II : Four g of propyne reacts with $$ NaNH_2 \text{ to liberate } NH_3$$ gas which occupies 224 mL at STP. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 57
The compounds which give positive Fehling's test are :
(A).
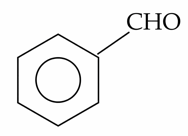
(B).
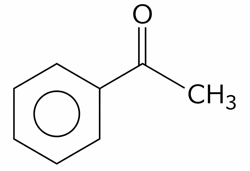
(C). $$ HOCH_{2} - CO - (CHOH)_{3}-CH_{2}-OH $$
(D). $$CH_{3}-CHO$$
(E).
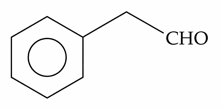
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 58
Which of the following electrolyte can be used to obtain $$H_2 S_2 O_8 $$ by the process of electrolysis?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 59
Given below are two statements : Statement I : $$ CH_3 - O - CH_2 - Cl$$ will undergo $$S_N 1$$ reaction though it is a
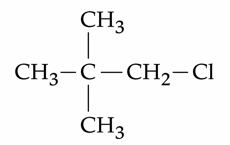
primary halide. Statement II :
will not undergo $$S_N 2$$ reaction very easily though it is a primary halide. In the light of the above statements,
choose the most appropriate answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 60
Which of the following acids is a vitamin?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 61
Match List-I with List-II.
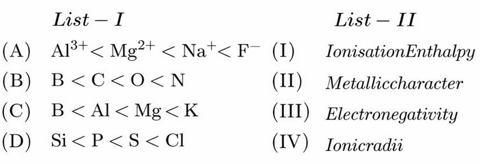
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 62
Which of the following statement is not true for radioactive decay?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 63
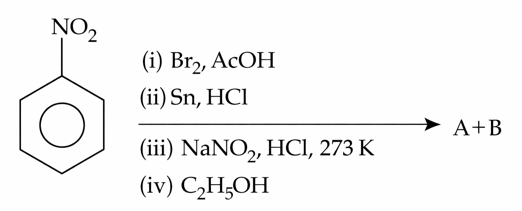
The products formed in the following reaction sequence are :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 64
How many different stereoisomers are possible for the given molecule?
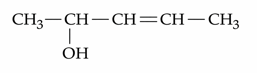
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 65
A vessel at 1000 K contains $$CO_2$$ with a pressure of 0.5 atm . Some of $$CO_2$$ is converted into CO on addition of graphite. If total pressure at equilibrium is 0.8 atm , then Kp is :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 66
A solution of aluminium chloride is electrolysed for 30 minutes using a current of 2 A . The amount of the aluminium deposited at the cathode is [Given : molar mass of aluminium and chlorine are 27g $$mol^{-1} $$ and 35.5 g $$mol^{-1}$$ respectively. Faraday constant $$=96500C mol^{-1}$$ ]
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 67
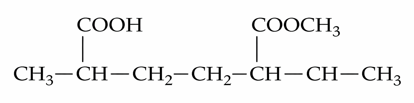
The IUPAC name of the following compound is :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 68
In which of the following complexes the CFSE, $$\triangle_o$$ will be equal to zero?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 69
Arrange the following solutions in order of their increasing boiling points. (i) $$10^{-4} M$$ $$NaCl$$ (ii) $$10^{-4}M$$ $$Urea$$ (iii) $$10^{-3} M$$ $$NaCl$$ (iv) $$10^{-2} M$$ $$NaCl$$
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 70
From the magnetic behaviour of $$[NiCl_4]^{2-} \text{(paramagnetic) and } [Ni(CO)_4] $$ (diamagnetic), choose the correct geometry and oxidation state.
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 71
The number of molecules/ions that show linear geometry among the following is ______
$$\mathrm{SO_2}$$, $$\ \mathrm{BeCl_2}$$, $$\ \mathrm{CO_2}$$, $$\ \mathrm{N_3^-}$$, $$\ \mathrm{NO_2}$$, $$\ \mathrm{F_2O}$$, $$\ \mathrm{XeF_2}$$, $$\ \mathrm{NO_2^+}$$, $$\ \mathrm{I_3^-}$$, $$\ \mathrm{O_3}$$
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 72
$$ A \rightarrow B $$ The molecule A changes into its isomeric form B by following a first order kinetics at a temperature
of 1000 K . If the energy barrier with respect to reactant energy for such isomeric transformation is $$191.48kJ$$ $$Mol^{-1}$$ and the frequency factor is $$10^{20},$$ the time required for 50% molecules of A to become B is
_________ picoseconds (nearest integer). $$[R=8.314 JK^{-1} mol^{-1}]$$
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 73
Consider the following sequence of reactions :
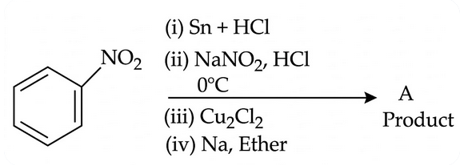
Molar mass of the product formed (A) is ______$$gmol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 74
Some $$CO_2$$ gas was kept in a sealed container at a pressure of 1 atm and at 273 K . This entire amount of $$CO_2$$ gas was later passed through an aqueous solution of $$Ca(OH)_2$$. The excess unreacted $$Ca(OH)_2$$ was later neutralized with 0.1 M of 40 mL HCl . If the volume of the sealed container of $$CO_2$$ was $$x$$, then $$x$$ is ________$$cm^{3}$$ (nearest integer). [Given : The entire amount of $$CO_2 (g)$$reacted with exactly half the initial amount of $$Ca(OH)_2$$ present in the aqueous solution.]
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Chemistry - Question 75
In Carius method for estimation of halogens, 180 mg of an organic compound produced 143.5 mg of AgCl . The percentage composition of chlorine in the compound is _______ \%. (Given : molar mass in $$gmol^{-1} \text{ of } Ag: 108, Cl: 35.5$$)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




