NTA JEE Mains 21st Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 61
Given below are two statements:
Statement I: The correct order in terms of bond dissociation enthalpy is $$Cl_{2} > Br_{2} > F_{2} > I_{2}$$.
Statement II : The correct trend in the covalent character of the metal halides is $$[SnCl_{4} > SnCl_{2}]$$, $$[PbCl_{4}> PbCl_{2}]$$, and $$[UF_{4} > UF_{6}]$$.
In The light oh the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 62
Consider the following spectral lines for atomic hydrogen :
A. First line of Paschen series
B. Second line of Balmer series
C. Third line of Paschen series
D. Fourth line of Bracket series
The correct arrangement of the above lines in ascending order of energy is :
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 63
Match List - I with List - II.
%20slot%202.png)
Choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 64
Given below are some of the statements about Mn and $$Mn_{2}O_{7}$$. Identify the correct statements.
A. Mn forms the oxide $$Mn_{2}O_{7}$$, in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn.
C. $$Mn_{2}O_{7}$$ is an ionic oxide.
D. The structure of $$Mn_{2}O_{7}$$ consists of one bridged oxygen.
Choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 65
Decompasition of A is a first order reaction at T(K) and is given by $$A(g) \rightarrow B(g)+C(g)$$.
In a closed 1 L vessel, 1 bar A(g) is allowed to decompose at T(K). After 100 minutes, the total pressure was 1.5bar. What is the rate constant $$(in min^{-1})$$ of the reaction ? (log 2 = 0.3)
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 66
The correct increasing order of C - H(A), C - 0 (B), C = O(C) and C = N (D) bonds in terms of covalent bond length is :
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 67
On heating a mixture of common salt and $$K_{2}Cr_{2}O_{7}$$ in equal amount along with concentrated $$H_{2}SO_{4}$$ in a test tube, a gas is evolved. Formula of the gas evolved and oxidation State of the central metal atom in the gas respectively are:
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 68
Given below are two statements:
Statement I: The correct order in terms of atomic/ionic radii is $$Al >Mg > Mg^{2+} >Al^{3+}$$
Statement II: The correct order in terms of the magnitude of electron gain enthalpy is Cl > Br >S >O.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 69
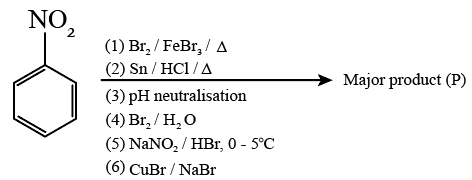
Consider the above sequence of reactions. The number of bromine atom(s) in the final product (P) will be:
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 70
The correct statements are :
A. Activation energy for enzyme catalysed hydrolysis of sucrose is lower than that of acid catalysed hydrolysis.
B. During denaturation, secondary and tertiary structures of a protein are destroyed but primary structure remains intact.
C. Nucleotides are joined together by glycosidic linkage between $$C_{1}$$ and $$C_{4}$$ carbons of the pentose sugar.
D. Quaternary structure of proteins represents overall folding of the polypeptide chain.
Choose the correct answer from the options given below :


