NTA JEE Mains 21st Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 51
The correct order of reactivity of the following benzyl halides towards reaction with KCN is:
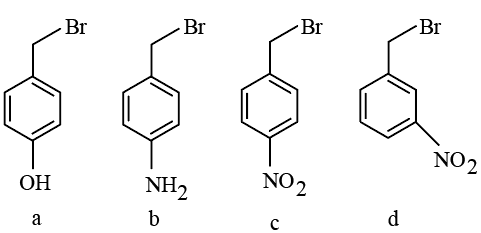
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 52
Given below are four compounds :
(a) n-propyl choride
(b) iso-propyl chloride
(c) sec-butyl chloride
(d) neo-pentyl chloride
Percentage of carbon in the one which exhibits optical isomerism is:
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 53
By usual analysis, 1.00 g of compow1d (X) gave 1.79 g of magnesium pyrophosphate. The percentage of phosphorus in compound (X) is: (nearest integer)
(Given, molar mass in $$gmol^{-1}$$ : 0 = 16, Mg = 24, P = 31 )
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 54
Given below are two statements:
Statement I : Crystal Field Stabilization Energy (CFSE) of $$\left[Cr\left( H_{2}O \right)_{6} \right]^{2+}$$ is greater than that of $$\left[Mn\left( H_{2}O \right)_{6} \right]^{2+}$$.
Statement II: Potassium ferricyanide has a greater spin-only magnetic moment than sodium ferrocyanide.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 55
Aqueous HCI reacts with $$MnO_{2} \left(s\right)$$ to form $$MnCl_{2}\left(aq\right)$$, $$Cl_{2}\left(g\right)$$ and $$H_{2}O\left(l\right)$$. What is the weight (in g) of Cl_{2} liberated when 8.7 g of $$MnO_{2} \left(s\right)$$ is reacted with excess aqueous HCI solution ?
(Given Molar mass in g $$mol^{-1}$$ Mn = 55, Cl = 35.5, 0 = 16, H = l )
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 56
For a closed circuit Daniell cell, which of the following plots is the accurate one at a given temperature?
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 57
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 58
Consider the following data :
$$\Delta_f H^\ominus$$ (methane, g) = - X kJ $$mol^{-1}$$
Enthalpy of sublimation of graphite = Y kJ $$mol^{-1}$$
Dissociation enthalpy of $$H_{2}$$ = Zkj $$mol^{-1}$$
The bond enthalpy of C - H bond is given by :
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 59
The correct order of the rate of the reaction for the following reaction with respect to nucleophiles is:
$$CH_{3}Br + Nu^{\ominus} \rightarrow CH_{3}Nu+Br^{\ominus}$$
NTA JEE Mains 21st Jan 2026 Shift 2 - Question 60
Given below are two statements:
Statement I: Compound (X), shown below, dissolves in $$NaHCO_{3}$$ solution and has two chiral carbon atoms
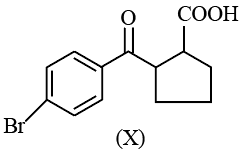
Statement II: Compound (Y), shown below, has two carbons with $$sp^{3}$$ hybridization, one carbon with $$sp^{2}$$ and one carbon with sp hybridization
%20slot%202.png)
In the light of the above statements, choose the correct answer from the options given below:


