NTA JEE Mains 2nd April Shift 2 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 46
In a Young's double slit experiment, the intensity at some point on the screen is found to be $$\frac{3}{4}$$ times of the maximum of the interference pattern. The path difference between the interfering waves at this point is $$\frac{\lambda}{x}$$ where $$\lambda$$ is wavelength of the incident light. The value of $$x$$ is _________.
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 47
Using Bohr's model, calculate the ratio of the magnetic fields generated due to the motion of the electrons in the 2nd and 4th orbits of hydrogen atom _________.
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 48
5 moles of unknown gas is heated at constant volume from 10 °C to 20 °C. The molar specific heat of this gas at constant pressure $$c_p = 8$$ cal/mol.°C and $$R = 8.36$$ J/mol.°C. The change in internal energy of the gas is _________ calorie.
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 49
If sunlight is focused on a paper using convex lens, it starts burning the paper in shortest time when the lens is kept at 30 cm above the paper. If the radius of curvature of the lens is 60 cm then the refractive index of the lens material is $$\frac{\alpha}{10}$$. The value of $$\alpha$$ is _________.
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 50
Moment of inertia about an axis $$AB$$ for a rod of mass 40 kg and length 3 m is same as that of a solid sphere of mass 10 kg and radius $$R$$ about an axis parallel to $$AB$$ axis with separation of 3 m as shown in figure. The value of $$R$$ is given as $$\sqrt{\frac{\alpha}{2}}$$. The value of $$\alpha$$ is _________.
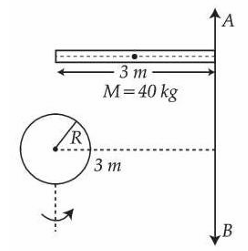
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 51
The ratio of mass percentage (w/w) of C : H in a hydrocarbon is 12 : 1. It has two carbon atoms. The weight (in g) of CO$$_2$$(g) formed when 3.38 g of this hydrocarbon is completely burnt in oxygen is : (Given: Molar mass in g mol$$^{-1}$$ C : 12, H : 1, O : 16)
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 52
The first and second ionization constants of a weak dibasic acid $$H_2A$$ are $$8.1 \times 10^{-8}$$ and $$1.0 \times 10^{-13}$$ respectively. 0.1 mol of $$H_2A$$ was dissolved in 1 L of 0.1 M HCl solution. The concentration of $$HA^-$$ in the resultant solution is :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 53
$$SF_4$$ is isostructural with :
A. $$BrF_4^\ominus$$
B. $$CH_4$$
C. $$IF_4^\oplus$$
D. $$XeF_4$$
E. $$XeO_2F_2$$
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 54
Gas 'A' undergoes change from state 'X' to state 'Y'. In this process, the heat absorbed and work done by the gas is 10 J and 18 J respectively. Now gas is brought back to state 'X' by another process during which 6 J of heat is evolved. In the reverse process of 'Y' to 'X' :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 55
Solution A is prepared by dissolving 1 g of a protein (molar mass = 50000 g mol$$^{-1}$$) in 0.5 L of water at 300 K. Its osmotic pressure is $$x$$ bar. Solution B is made by dissolving 2 g of same protein in 1 L of water at 300 K. Osmotic pressure of solution B is $$y$$ bar. Entire solution of A is mixed with entire solution of B at same temperature. The osmotic pressure of resultant solution is $$z$$ bar. $$x$$, $$y$$ and $$z$$ respectively are :
($$R = 0.083$$ L bar mol$$^{-1}$$ K$$^{-1}$$)
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 56
At 25°C, 20.0 mL of 0.2 M weak monoprotic acid HX is titrated against 0.2 M NaOH. The pH of the solution (a) at the start of the titration (when NaOH has not been added) and (b) when 10 mL of NaOH is added respectively, are :
Given: $$K_a = 5 \times 10^{-4}$$,
$$pK_a = 3.3$$,
$$\alpha \ll 1$$
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 57
Consider the reaction $$aX \to bY$$, for which the rate constant at 30°C is $$1 \times 10^{-3}$$ mol$$^{-1}$$ L s$$^{-1}$$. Which of the following statements are true?
A. When concentration of 'X' is increased to four times, the rate of reaction becomes 16 times.
B. The reaction is a second order reaction.
C. The half-life period is independent of the concentration of X.
D. Decomposition of $$N_2O_5$$ is an example of the above reaction.
E.
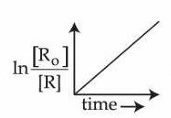
is valid for the above reaction.
Choose the correct answer from the option given below:
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 58
The correct set that contains all kinds (basic, acidic, amphoteric and neutral) of oxides is :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 59
Given below are two statements :
Statement I : The second ionization enthalpy of B, Al and Ga is in the order of $$B > Al > Ga$$.
Statement II : The correct order in terms of first ionization enthalpy is $$Si < Ge < Pb < Sn$$.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 60
Given below are two statements :
Statement I : Among Zn, Mn, Sc and Cu, the energy required to remove the third valence electron is highest for Zn and lowest for Sc.
Statement II : The correct order of the following complexes in terms of CFSE is $$[Co(H_2O)_6]^{2+} < [Co(H_2O)_6]^{3+} < [Co(en)_3]^{3+}$$.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 61
Which of the following complexes will show coordination isomerism?
A. $$[Ag(NH_3)_2][Ag(CN)_2]$$
B. $$[Co(NH_3)_6][Cr(CN)_6]$$
C. $$[Co(NH_3)_6][Co(CN)_6]$$
D. $$[Fe(NH_3)_6][Co(CN)_6]$$
E. $$[Co(NH_3)_6][Fe(CN)_6]$$
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 62
Complete combustion of $$X$$ g of an organic compound gave 0.25 g of CO$$_2$$ and 0.12 g of H$$_2$$O. If the % of carbon is 25% and of hydrogen is 4.89%, then $$X = $$ _____ $$\times 10^{-3}$$ g. (Nearest integer) (Molar mass of C, H and O are 12, 1 and 16 g mol$$^{-1}$$ respectively.)
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 63
Given below are two statements :
Statement I : In
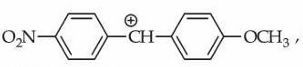
the carbocation is stabilised by +R effect of $$-OCH_3$$ group.
Statement II : In
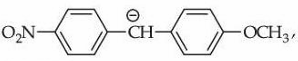
the carbanion is stabilised by $$-R$$ effect of $$-NO_2$$ group.
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 64
The compound (X) on
(i) on heating in the presence of anhydrous AlCl$$_3$$ and HCl gas gives 2,4-dimethyl pentane
(ii) aromatization gives toluene and
(iii) cyclisation gives methyl cyclohexane
The correct name of compound (X) is :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 65
Correct statements regarding alkyl halides $$(R-X)$$ among the following are :
A. Alcohol being less polar solvent favours elimination with alcoholic KOH favours elimination reaction with $$R - X.$$
B. Order of reactivity towards $$S_N1$$ is $$C_6H_5-CH_2-Cl > C_6H_5-CHCl-C_6H_5$$.
C. Non substituted aryl halides exhibit properties similar to alkyl halides.
D. Vinyl chloride is example of haloalkene and allyl chloride is example of haloalkyne.
E. $$R-Cl$$ can be prepared by reacting $$R-OH$$ with $$SOCl_2$$ but $$Ar-Cl$$ cannot be prepared by reacting $$Ar-OH$$ with $$SOCl_2$$.
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 66
An organic compound "x" where molar ratio of C, O and H are equal, on treatment with 50% KOH under reflux followed by acidification produced "y". The most likely structure of "y" is :
[Molar mass of 'x' is 58 g mol$$^{-1}$$]
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 67
A molecule (X) with the following structure under mild acidic condition is hydrolysed to produce (Y) and (Z). Identify the correct statements about (Y) and (Z).
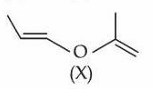
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$$_3$$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 68
Identify compounds A and E in the following reaction sequence.
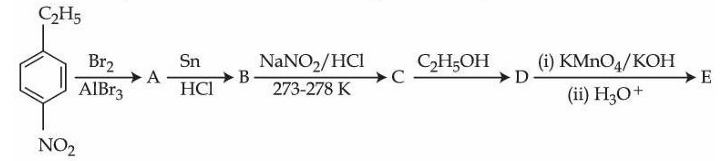
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 69
Identify the correct pair having amino acid (A) and the hormone (B) that is iodinated derivative of the amino acid (A).
(T and Y represent one letter code for amino acids)
Amino acid (A) Hormone (B)
T Insulin
T Thyroxine
Y Thyroxine
Y Insulin
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 70
Among $$Fe^{2+}$$, $$Fe^{3+}$$, $$Cr^{2+}$$ and $$Zn^{2+}$$, the ion that shows positive borax bead test and with highest ionisation enthalpy is :
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 71
The surface of sodium metal is irradiated with radiation of wavelength $$x$$ nm. The kinetic energy of ejected electrons is $$2.8 \times 10^{-20}$$ J. The work function of sodium is 2.3 eV. The value of $$x$$ is _____ $$\times 10^2$$ nm. (Nearest integer) (Given: $$h = 6.6 \times 10^{-34}$$ J s; $$1$$ eV $$= 1.6 \times 10^{-19}$$ J; $$c = 3.0 \times 10^8$$ m s$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 72
Consider the following gas phase reaction being carried out in a closed vessel at 25°C.
$$2A(g) \to 4B(g) + C(g)$$

The pressure of $$C(g)$$ at 30 minutes time interval would be _____ mm Hg. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 73
Consider the following two half-cell reactions along with the standard reduction potential given :
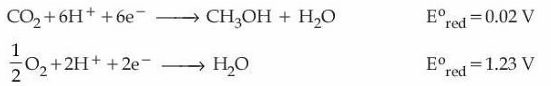
A fuel cell was set up using the above two reactions such that the cell operates under the standard condition of 1 bar pressure and 298 K temperature. The fuel cell works with 80% efficiency. If the work derived from the cell using 1 mol of CH$$_3$$OH is used to compress an ideal gas isothermally against a constant pressure of 1 kPa, then the change in the volume of the gas, $$\Delta V = $$ _____ m$$^3$$. (nearest integer)
Given: $$F = 96500$$ C mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 74
Number of paramagnetic ions among the following d- and f-block metal ions is _________.
$$Mn^{2+}$$, $$Cu^{2+}$$, $$Zn^{2+}$$, $$Yb^{2+}$$, $$Sc^{3+}$$, $$La^{3+}$$, $$Gd^{3+}$$, $$Lu^{3+}$$, $$Ti^{4+}$$, $$Ce^{4+}$$
(Atomic number of Mn = 25, Cu = 29, Zn = 30, Yb = 70, Sc = 21, La = 57, Gd = 64, Lu = 71, Ti = 22, Ce = 58)
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Chemistry - Question 75
Consider the following reactions sequence.
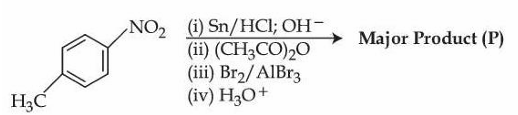
When the product (P) is subjected to Carius analysis using AgNO$$_3$$, 1.0 g of the product (P) will produce _____ g of the precipitate of AgBr. (Nearest Integer)
(Given: molar mass in g mol$$^{-1}$$ C: 12, H: 1, O: 16, N: 14, Br: 80, Ag: 108)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




