NTA JEE Mains 1st feb 2023 Shift 2
For the following questions answer them individually
NTA JEE Mains 1st feb 2023 Shift 2 - Question 41
Given below are two statements:
Statement I: Sulphanilic acid gives esterification test for carboxyl group.
Statement II: Sulphanilic acid gives red colour in Lassigne's test for extra element detection.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st feb 2023 Shift 2 - Question 42
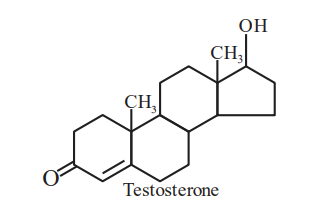
Testosterone, which is a steroidal hormone, has the following structure. The total number of asymmetric carbon atom/s in testosterone is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 1st feb 2023 Shift 2 - Question 43
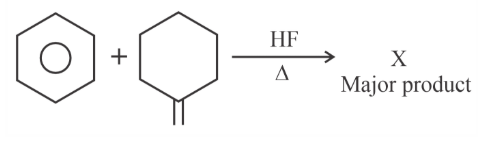
'X' is:
NTA JEE Mains 1st feb 2023 Shift 2 - Question 44
The industrial activity held least responsible for global warming is :
NTA JEE Mains 1st feb 2023 Shift 2 - Question 45
A metal M crystallizes into two lattices :- face centred cubic (fcc) and body centred cubic (bcc) with unit cell edge length of $$2.0$$ and $$2.5$$ Å respectively. The ratio of densities of lattices fcc to bcc for the metal M is ______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st feb 2023 Shift 2 - Question 46
20% of acetic acid is dissociated when its 5 g is added to 500 mL of water. The depression in freezing point of such water is ______ $$\times 10^{-3}°C$$. Atomic mass of C, H and O are 12, 1 and 16 a.m.u. respectively.
[Given : Molal depression constant and density of water are $$1.86 \text{ K kg mol}^{-1}$$ and $$1 \text{ g cm}^{-3}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Mains 1st feb 2023 Shift 2 - Question 47
$$1 \times 10^{-5}$$ M $$AgNO_3$$ is added to 1 L of saturated solution of AgBr. The conductivity of this solution at 298 K is ______ $$\times 10^{-8} \text{ S m}^{-1}$$.
[Given : $$K_{sp}(AgBr) = 4.9 \times 10^{-13}$$ at 298 K, $$\lambda^0_{Ag^+} = 6 \times 10^{-3} \text{ Sm}^2 \text{ mol}^{-1}$$, $$\lambda^0_{Br^-} = 8 \times 10^{-3} \text{ Sm}^2 \text{ mol}^{-1}$$, $$\lambda^0_{NO_3^-} = 7 \times 10^{-3} \text{ Sm}^2 \text{ mol}^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 1st feb 2023 Shift 2 - Question 48
The graph which represents the following reaction is:
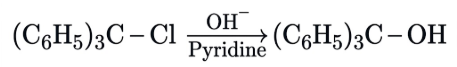
NTA JEE Mains 1st feb 2023 Shift 2 - Question 49
$$A \rightarrow B$$
The above reaction is of zero order. Half life of this reaction is 50 min. The time taken for the concentration of A to reduce to one-fourth of its initial value is ______ min. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st feb 2023 Shift 2 - Question 50
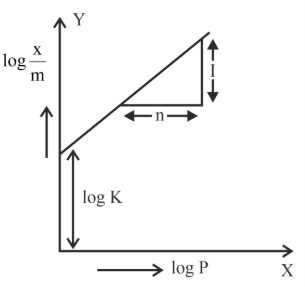
In figure, a straight line is given for Freundrich Adsorption $$(y = 3x + 2.505)$$. The value of $$\frac{1}{n}$$ and $$\log K$$ are respectively.