NTA JEE Mains 04th April 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 31
Choose the Incorrect Statement about Dalton's Atomic Theory
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 32
The correct order of the first ionization enthalpy is
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 33
Given below are two statements: Statement I: The correct order of first ionization enthalpy values of Li, Na, F and Cl is $$Na < Li < Cl < F$$. Statement II: The correct order of negative electron gain enthalpy values of Li, Na, F and Cl is $$Na < Li < F < Cl$$. In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 34
The correct statement/s about Hydrogen bonding is/are: A. Hydrogen bonding exists when H is covalently bonded to the highly electronegative atom. B. Intermolecular H bonding is present in o-nitrophenol. C. Intramolecular H bonding is present in HF. D. The magnitude of H bonding depends on the physical state of the compound. E. H-bonding has powerful effect on the structure and properties of compounds. Choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 35
The number of species from the following that have pyramidal geometry around the central atom is _______. $$S_2O_3^{2-}$$, $$SO_4^{2-}$$, $$SO_3^{2-}$$, $$S_2O_7^{2-}$$
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 36
The equilibrium constant for the reaction $$SO_3(g) \rightleftharpoons SO_2(g) + \frac{1}{2}O_2(g)$$ is $$K_c = 4.9 \times 10^{-2}$$. The value of $$K_c$$ for the reaction given below is $$2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)$$:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 37
Correct order of stability of carbanion is
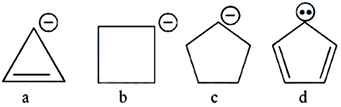
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 38
Common name of Benzene-1,2-diol is:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 39
The adsorbent used in adsorption chromatography is/are: A. silica gel, B. alumina, C. quick lime, D. magnesia. Choose the most appropriate answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 41
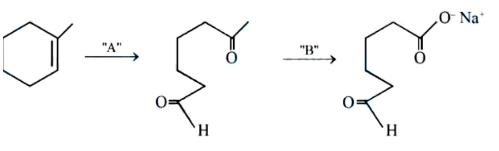
In the above chemical reaction sequence "A" and "B" respectively are:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 42
For a strong electrolyte, a plot of molar conductivity against (concentration)$$^{1/2}$$ is a straight line, with a negative slope, the correct unit for the slope is:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 43
Fuel cell, using hydrogen and oxygen as fuels: A. has been used in spaceship, B. has an efficiency of 40% to produce electricity, C. uses aluminum as catalysts, D. is eco-friendly, E. is actually a type of Galvanic cell only. Choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 44
When $$MnO_2$$ and $$H_2SO_4$$ is added to a salt (A), the greenish yellow gas liberated as salt (A) is:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 45
A first row transition metal in its +2 oxidation state has a spin-only magnetic moment value of $$3.86$$ BM. The atomic number of the metal is:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 46
If an iron (III) complex with the formula $$[Fe(NH_3)_x(CN)_y]^-$$ has no electron in its $$e_g$$ orbital, then the value of $$x + y$$ is:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 47
The number of unpaired d-electrons in $$[Co(H_2O)_6]^{3+}$$ is:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 48
$$CH_3-CH_2-CH_2-Br + NaOH \xrightarrow{C_2H_5OH}$$ Product 'A'.
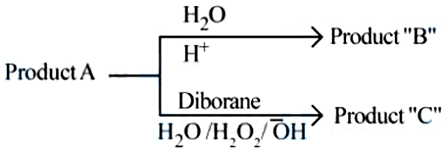
Consider the above reactions, identify product $$B$$ and product $$C$$.
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 49
Find out the major product formed from the following reaction. [Me: $$-CH_3$$]
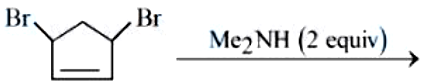
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 50
Match List I with List II:
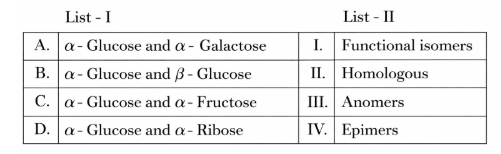
Choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 51
The maximum number of orbitals which can be identified with $$n = 4$$ and $$m_l = 0$$ is ______
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 52
Number of compounds/species from the following with non-zero dipole moment is ______. $$BeCl_2$$, $$BCl_3$$, $$NF_3$$, $$XeF_4$$, $$CCl_4$$, $$H_2O$$, $$H_2S$$, $$HBr$$, $$CO_2$$, $$H_2$$, $$HCl$$
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 53
Three moles of an ideal gas are compressed isothermally from $$60$$ L to $$20$$ L using constant pressure of $$5$$ atm. Heat exchange $$Q$$ for the compression is $$-$$ ______ Lit. atm.
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 54
The total number of 'sigma' and 'Pi' bonds in 2-oxohex-4-ynoic acid is ______
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 55
$$2.7$$ kg of each of water and acetic acid are mixed. The freezing point of the solution will be $$-x°C$$. Consider the acetic acid does not dimerise in water, nor dissociates in water. $$x =$$ ______ (nearest integer) [Given: Molar mass of water $$= 18$$ g mol$$^{-1}$$, acetic acid $$= 60$$ g mol$$^{-1}$$, $$K_f$$ H$$_2$$O: $$1.86$$ K kg mol$$^{-1}$$, $$K_f$$ acetic acid: $$3.90$$ K kg mol$$^{-1}$$, freezing point: H$$_2$$O $$= 273$$ K, acetic acid $$= 290$$ K]
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 56
Consider the following reaction, the rate expression of which is given below:
$$A + B \rightarrow C$$, rate$$ = k[A]^{1/2}[B]^{1/2}$$.
The reaction is initiated by taking concentration of $$1$$M of $$A$$ and $$B$$ each. If the rate constant $$(k)$$ is $$4.6 \times 10^{-2}$$ s$$^{-1}$$, then the time taken for $$A$$ to become $$0.1$$M is ______ sec. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 57
A first row transition metal with highest enthalpy of atomisation, upon reaction with oxygen at high temperature forms oxides of formula $$M_2O_n$$ (where $$n = 3, 4, 5$$). The 'spin-only' magnetic moment value of the amphoteric oxide from the above oxides is ______ BM (nearest integer). (Given atomic number: Sc: 21, Ti: 22, V: 23, Cr: 24, Mn: 25, Fe: 26, Co: 27, Ni: 28, Cu: 29, Zn: 30)
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 58
Phthalimide is made to undergo following sequence of reactions:
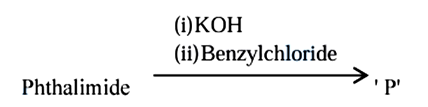
Total number of $$\pi$$ bonds present in product 'P' is/are ______
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 59
From $$6.55$$ g of aniline, the maximum amount of acetanilide that can be prepared will be ______ $$\times 10^{-1}$$ g.
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 2 - Chemistry - Question 60
Vanillin compound obtained from vanilla beans, has total sum of oxygen atoms and $$\pi$$ electrons is ______
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
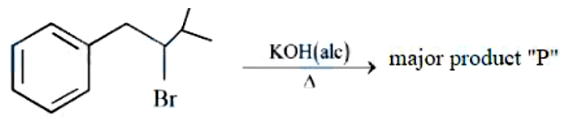
.webp)




