NTA JEE Mains 1st Feb 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 51
10 mL of gaseous hydrocarbon on combustion gives 40 mL of $$CO_2(g)$$ and 50 mL of water vapour. Total number of carbon and hydrogen atoms in the hydrocarbon is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 52
For a certain reaction at 300 K, K = 10, then $$\Delta G°$$ for the same reaction is - ______ $$\times$$ $$10^{-1}$$ $$kJ mol^{-1}$$. (Given $$R = 8.314 \text{ J K}^{-1} \text{mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 53
Following Kjeldahl's method, 1 g of organic compound released ammonia, that neutralised 10 mL of $$2M$$ $$H_2SO_4$$. The percentage of nitrogen in the compound is ______ %.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 54
Total number of isomeric compounds (including stereoisomers) formed by monochlorination of 2-methylbutane is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 55
Mass of ethylene glycol (antifreeze) to be added to 18.6 kg of water to protect the freezing point at $$-24°C$$ is ______ kg (Molar mass in g mol$$^{-1}$$ for ethylene glycol 62, $$K_f$$ of water = $$1.86 \text{ K kg mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 56
The amount of electricity in Coulomb required for the oxidation of 1 mol of $$H_2O$$ to $$O_2$$ is ______ $$\times 10^5$$ C.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 57
Consider the following redox reaction: $$MnO_4^- + H^+ + H_2C_2O_4 \rightleftharpoons Mn^{2+} + H_2O + CO_2$$
The standard reduction potentials are given as below:
$$E^0_{MnO_4^-/Mn^{2+}} = +1.51$$ V; $$E^0_{CO_2/H_2C_2O_4} = -0.49$$ V
If the equilibrium constant of the above reaction is given as $$K_{eq} = 10^x$$, then the value of $$x$$ = ______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 58
The following data were obtained during the first order thermal decomposition of a gas A at constant volume:
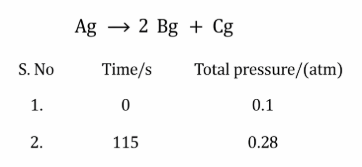
The rate constant of the reaction is ______ $$\times 10^{-2} \text{ s}^{-1}$$ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 59
Number of compounds which give reaction with Hinsberg's reagent is ______.
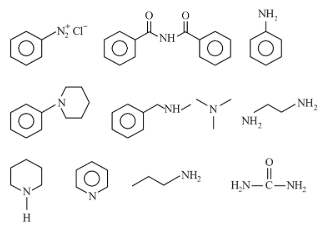
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 60
The number of tripeptides formed by three different amino acids using each amino acid once is ______.
789
456
123
0.-
Clear All


