NTA JEE Mains 1st Feb 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 41
Lassaigne's test is used for detection of:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 42
In the given reactions identify A and B.
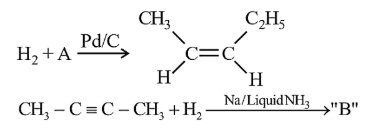
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 43
The strongest reducing agent among the following is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 44
The transition metal having highest 3rd ionisation enthalpy is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 45
Which of the following compounds show colour due to d-d transition?
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 46
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: In aqueous solutions $$Cr^{2+}$$ is reducing while $$Mn^{3+}$$ is oxidising in nature.
Reason R: Extra stability to half filled electronic configuration is observed than incompletely filled electronic configuration.
In the light of the above statement, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 47
Given below are two statements:
Statement (I): Dimethyl glyoxime forms a six membered covalent chelate when treated with $$NiCl_2$$ solution in presence of $$NH_4OH$$.
Statement (II): Prussian blue precipitate contains iron both in +2 and +3 oxidation states.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 48
$$[Co(NH_3)_6]^{3+}$$ and $$[CoF_6]^{3-}$$ are respectively known as:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 49
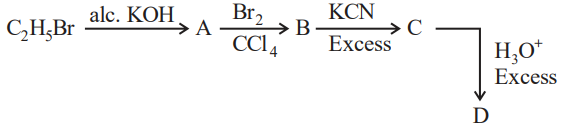
Acid D formed in above reaction is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Question 50
Match List - I with List - II.

Choose the correct answer from the options given below:


