NTA JEE Mains 1st Feb 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 31
According to the wave-particle duality of matter by de-Broglie, which of the following graph plot presents most appropriate relationship between wavelength of electron $$\lambda$$ and momentum of electron $$p$$? (Four graphs are shown: (1) $$\lambda$$ vs $$p$$ showing a rectangular hyperbola, (2) $$p$$ vs $$\lambda$$ showing a rectangular hyperbola, (3) $$\lambda$$ vs $$1/p$$ showing a straight line through origin, (4) $$\lambda$$ vs $$p$$ showing a straight line with negative slope)
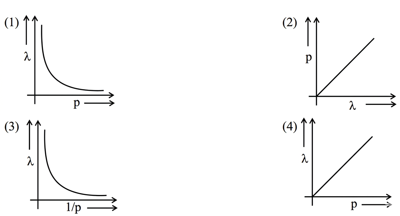
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 32
In case of isoelectronic species the size of $$F^-$$, $$Ne$$ and $$Na^+$$ is affected by:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 33
Arrange the bonds in order of increasing ionic character in the molecules: $$LiF$$, $$K_2O$$, $$N_2$$, $$SO_2$$ and $$ClF_3$$.
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 34
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: $$PH_3$$ has lower boiling point than $$NH_3$$. Reason R: In liquid state $$NH_3$$ molecules are associated through Vander Waal's forces, but $$PH_3$$ molecules are associated through hydrogen bonding. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 35
Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following :
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 36
Which of the following reactions are disproportionation reactions? (1) $$Cu^+ \rightarrow Cu^{2+} + Cu$$ (2) $$3MnO_4^{2-} + 4H^+ \rightarrow 2MnO_4^- + MnO_2 + 2H_2O$$ (3) $$2KMnO_4 \rightarrow K_2MnO_4 + MnO_2 + O_2$$ (4) $$2MnO_4^- + 3Mn^{2+} + 2H_2O \rightarrow 5MnO_2 + 4H^+$$. Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 37
In acidic medium, $$K_2Cr_2O_7$$ shows oxidising action as represented in the half reaction $$Cr_2O_7^{2-} + XH^+ + Ye^- \rightarrow 2A + ZH_2O$$. X, Y, Z and A are respectively:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 38
Given below are two statements: Statement (I): Potassium hydrogen phthalate is a primary standard for standardisation of sodium hydroxide solution. Statement (II): In this titration phenolphthalein can be used as indicator. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 39
Given below are two statements: Statement (I): Aminobenzene and aniline are same organic compounds. Statement (II): Aminobenzene and aniline are different organic compounds. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 40
Ionic reactions with organic compounds proceed through: (A) Homolytic bond cleavage (B) Heterolytic bond cleavage (C) Free radical formation (D) Primary free radical (E) Secondary free radical. Choose the correct answer from the options given below:


