NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 31
According to the wave-particle duality of matter by de-Broglie, which of the following graph plot presents most appropriate relationship between wavelength of electron $$\lambda$$ and momentum of electron $$p$$? (Four graphs are shown: (1) $$\lambda$$ vs $$p$$ showing a rectangular hyperbola, (2) $$p$$ vs $$\lambda$$ showing a rectangular hyperbola, (3) $$\lambda$$ vs $$1/p$$ showing a straight line through origin, (4) $$\lambda$$ vs $$p$$ showing a straight line with negative slope)
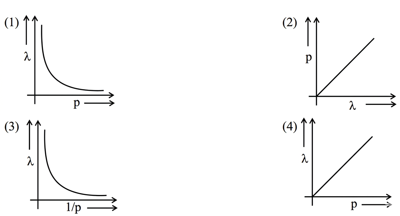
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 32
In case of isoelectronic species the size of $$F^-$$, $$Ne$$ and $$Na^+$$ is affected by:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 33
Arrange the bonds in order of increasing ionic character in the molecules: $$LiF$$, $$K_2O$$, $$N_2$$, $$SO_2$$ and $$ClF_3$$.
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 34
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: $$PH_3$$ has lower boiling point than $$NH_3$$. Reason R: In liquid state $$NH_3$$ molecules are associated through Vander Waal's forces, but $$PH_3$$ molecules are associated through hydrogen bonding. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 35
Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following :
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 36
Which of the following reactions are disproportionation reactions? (1) $$Cu^+ \rightarrow Cu^{2+} + Cu$$ (2) $$3MnO_4^{2-} + 4H^+ \rightarrow 2MnO_4^- + MnO_2 + 2H_2O$$ (3) $$2KMnO_4 \rightarrow K_2MnO_4 + MnO_2 + O_2$$ (4) $$2MnO_4^- + 3Mn^{2+} + 2H_2O \rightarrow 5MnO_2 + 4H^+$$. Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 37
In acidic medium, $$K_2Cr_2O_7$$ shows oxidising action as represented in the half reaction $$Cr_2O_7^{2-} + XH^+ + Ye^- \rightarrow 2A + ZH_2O$$. X, Y, Z and A are respectively:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 38
Given below are two statements: Statement (I): Potassium hydrogen phthalate is a primary standard for standardisation of sodium hydroxide solution. Statement (II): In this titration phenolphthalein can be used as indicator. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 39
Given below are two statements: Statement (I): Aminobenzene and aniline are same organic compounds. Statement (II): Aminobenzene and aniline are different organic compounds. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 40
Ionic reactions with organic compounds proceed through: (A) Homolytic bond cleavage (B) Heterolytic bond cleavage (C) Free radical formation (D) Primary free radical (E) Secondary free radical. Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 41
In Kjeldahl's method for estimation of nitrogen, $$CuSO_4$$ acts as :
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 42
Which of the following compound will most easily be attacked by an electrophile?
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 43
We have three aqueous solutions of NaCl labelled as 'A', 'B' and 'C' with concentration $$0.1$$ M, $$0.01$$ M and $$0.001$$ M, respectively. The value of van't Hoff factor $$i$$ for these solutions will be in the order:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 44
Which of the following complex is homoleptic?
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 45
Given below are two statements: Statement (I): A solution of $$[Ni(H_2O)_6]^{2+}$$ is green in colour. Statement (II): A solution of $$[Ni(CN)_4]^{2-}$$ is colourless. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 46
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: Haloalkanes react with KCN to form alkyl cyanides as a main product while with AgCN form isocyanide as the main product. Reason R: KCN and AgCN both are highly ionic compounds. In the light of the above statement, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 47
Identify A and B in the following sequence of reaction:
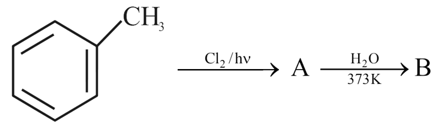
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 48
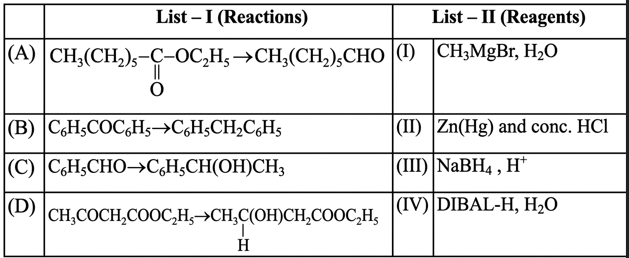
Choose the correct answer from options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 49
Given below are two statements: Statement (I): The $$-NH_2$$ group in Aniline is ortho and para directing and a powerful activating group. Statement (II): Aniline does not undergo Friedel-Craft's reaction (alkylation and acylation). In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 50
If one strand of a DNA has the sequence ATGCTTCA, sequence of the bases in complementary strand is:
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 51
Consider the following reaction: $$3PbCl_2 + 2(NH_4)_3PO_4 \rightarrow Pb_3(PO_4)_2 + 6NH_4Cl$$. If $$72$$ mmol $$PbCl_2$$ is mixed with $$50$$ mmol of $$(NH_4)_3PO_4$$, then amount of $$Pb_3(PO_4)_2$$ formed in mmol is (nearest integer):
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 52
Lowest oxidation number of an atom in a compound $$A_2B$$ is $$-2$$. The number of electrons in its valence shell is:
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 53
The number of molecules/ion/s having trigonal bipyramidal shape is: $$PF_5$$, $$BrF_5$$, $$PCl_5$$, $$[PtCl_4]^{2-}$$, $$BF_3$$, $$Fe(CO)_5$$
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 54
$$K_a$$ for $$CH_3COOH$$ is $$1.8 \times 10^{-5}$$ and $$K_b$$ for $$NH_4OH$$ is $$1.8 \times 10^{-5}$$. The pH of ammonium acetate solution will be:
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 55
Number of optical isomers possible for 2-chlorobutane is:
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 56
Total number of deactivating groups in aromatic electrophilic substitution reaction among the following is:
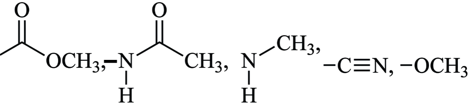
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 57
The potential for the given half cell at 298K is $$- x \times 10^{-2}$$ V.
$$2H^+_{(aq)} + 2e^- \rightarrow H_2(g)$$, $$[H^+] = 1$$ M, $$P_{H_2} = 2$$ atm.
(Given $$2.303\ RT/F = 0.06$$ V, $$\log 2 = 0.3$$). The value of $$x$$ is:
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 58
The ratio of $$\frac{^{14}C}{^{12}C}$$ in a piece of wood is $$\frac{1}{8}$$ part that of atmosphere. If half life of $$^{14}C$$ is $$5730$$ years, the age of wood sample is _____ years.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 59
Among the following oxides of p-block elements: $$Cl_2O_7$$, $$CO$$, $$PbO_2$$, $$N_2O$$, $$NO$$, $$Al_2O_3$$, $$SiO_2$$, $$N_2O_5$$, $$SnO_2$$, the number of amphoteric oxides is:
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 1 - Chemistry - Question 60
The number of white coloured salts among the following is: (A) $$SrSO_4$$ (B) $$MgNH_4PO_4$$ (C) $$BaCrO_4$$ (D) $$Mn(OH)_2$$ (E) $$PbSO_4$$ (F) $$PbCrO_4$$ (G) $$AgBr$$ (H) $$PbI_2$$ (I) $$CaC_2O_4$$ (J) $$Fe(OH)_2(CH_3COO)$$
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




