NTA JEE Main 9th April 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 9th April 2019 Shift 2 - Question 41
Molal depression constant for a solvent is 4.0 K kg mol$$^{-1}$$. The depression in the freezing point of the solvent for 0.03 mol kg$$^{-1}$$ solution of K$$_2$$SO$$_4$$ is: (Assume complete dissociation of the electrolyte)
NTA JEE Main 9th April 2019 Shift 2 - Question 42
A solution of Ni(NO$$_3$$)$$_2$$ is electrolyzed between platinum electrode 0.1 Faraday electricity. How many mole of Ni will be deposited at the cathode?
NTA JEE Main 9th April 2019 Shift 2 - Question 43
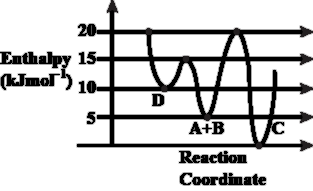
Consider the given plot of enthalpy of the following reaction between A and B.
A + B $$\rightarrow$$ C + D. Identify the incorrect statement.
NTA JEE Main 9th April 2019 Shift 2 - Question 44
10 mL of 1 mM surfactant solution forms a monolayer covering 0.24 cm$$^2$$ on a polar substrate. If the polar head is approximated as a cube, what is its edge length?
NTA JEE Main 9th April 2019 Shift 2 - Question 45
The one that is not a carbonate ore is:
NTA JEE Main 9th April 2019 Shift 2 - Question 46
Assertion: For the extraction of iron, haematite ore is used.
Reason: Haematite is a carbonate ore of iron.
NTA JEE Main 9th April 2019 Shift 2 - Question 47
HF has highest boiling point among hydrogen halides, because it has:
NTA JEE Main 9th April 2019 Shift 2 - Question 48
The maximum number of possible oxidation states of actinoids are shown by:
NTA JEE Main 9th April 2019 Shift 2 - Question 49
The maximum possible denticities of a ligand given below towards a common transition and inner-transition metal ion, respectively, are: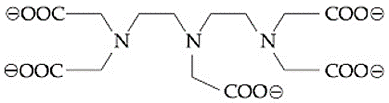
NTA JEE Main 9th April 2019 Shift 2 - Question 50
The correct statements among I to III are:
(I) Valence bond theory cannot explain the color exhibited by transition metal complexes.
(II) Valence bond theory can predict quantitatively the magnetic properties of transition metal complexes.
(III) Valence bond theory cannot distinguish ligands as weak and strong field ones.


