NTA JEE Main 9th April 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 9th April 2019 Shift 1 - Question 41
The major product of the following reaction is:
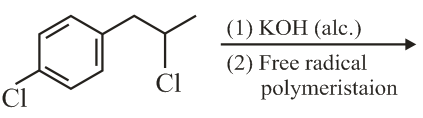
NTA JEE Main 9th April 2019 Shift 1 - Question 42
The major product of the following reaction is:
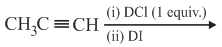
NTA JEE Main 9th April 2019 Shift 1 - Question 43
Excessive release of CO$$_2$$ into the atmosphere results in:
NTA JEE Main 9th April 2019 Shift 1 - Question 44
The osmotic pressure of a dilute solution of an ionic compound XY in water is four times that of a solution of 0.01 M BaCl$$_2$$ in water. Assuming complete dissociation of the given ionic compounds in water, the concentration of XY (in mol L$$^{-1}$$) in solution is:
NTA JEE Main 9th April 2019 Shift 1 - Question 45
Liquid M and liquid N form an ideal solution. The vapour pressures of pure liquids M and N are 450 and 700 mmHg, respectively, at the same temperature. Then correct statements is:
(x$$_M$$ = Mole fraction of 'M' in solution; x$$_N$$ = Mole fraction of 'N' in solution; y$$_M$$ = Mole fraction of 'M' in vapour phase; y$$_N$$ = Mole fraction of 'N' in vapour phase)
NTA JEE Main 9th April 2019 Shift 1 - Question 46
The standard Gibbs energy for the given cell reaction in kJ mol$$^{-1}$$ at 298 K is:
Zn(s) + Cu$$^{2+}$$(aq) $$\rightarrow$$ Zn$$^{2+}$$(aq) + Cu(s),
E$$^0$$ = 2 V at 298 K
(Faraday's constant, F = 96000 C mol$$^{-1}$$)
NTA JEE Main 9th April 2019 Shift 1 - Question 47
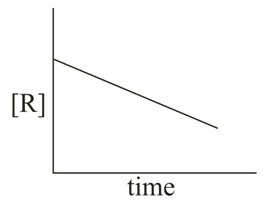
The given plots represent the variation of the concentration of a reactant R with time for two different reactions (i) and (ii). The respective orders of the reaction are: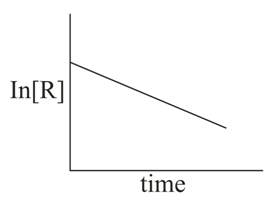
NTA JEE Main 9th April 2019 Shift 1 - Question 48
The aerosol is a kind of colloid in which:
NTA JEE Main 9th April 2019 Shift 1 - Question 49
The ore that contains the metal in the form of fluoride is known as which of the following?
NTA JEE Main 9th April 2019 Shift 1 - Question 50
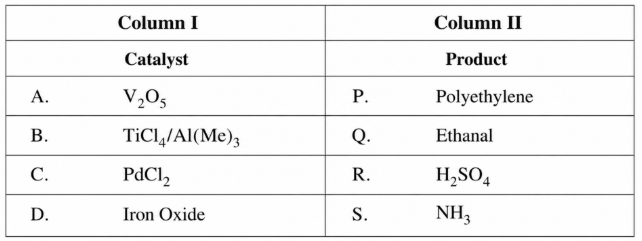
Match the catalysts (Column I) with products (Column II)