NTA JEE Main 9th April 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 9th April 2019 Shift 1 - Question 31
For a reaction, N$$_2$$(g) + 3H$$_2$$(g) $$\rightarrow$$ 2NH$$_3$$(g), identify di-hydrogen (H$$_2$$) as a limiting reagent in the following reaction mixtures.
NTA JEE Main 9th April 2019 Shift 1 - Question 32
For any given series of spectral lines of atomic hydrogen, let $$\Delta\bar{v} = \bar{v}_{max} - \bar{v}_{min}$$ be the difference in maximum and minimum wave number in cm$$^{-1}$$. The ratio $$\Delta\bar{v}_{Lyman}/\Delta\bar{v}_{Balmer}$$ is:
NTA JEE Main 9th April 2019 Shift 1 - Question 33
The element having the greatest difference between its first and second ionization energies, is:
NTA JEE Main 9th April 2019 Shift 1 - Question 34
Among the following, the molecule expected to be stabilized by anion formation is: C$$_2$$, O$$_2$$, NO, F$$_2$$
NTA JEE Main 9th April 2019 Shift 1 - Question 35
Consider the van der Waal's constants, a and b, for the following gases.

Which gas is expected to have the highest critical temperature?
NTA JEE Main 9th April 2019 Shift 1 - Question 36
Among the following, the set of parameters that represents path functions, is:
i) q + w
ii) q
iii) w
iv) H - TS
NTA JEE Main 9th April 2019 Shift 1 - Question 37
Magnesium powder burns in air to give:
NTA JEE Main 9th April 2019 Shift 1 - Question 38
C$$_{60}$$, an allotrope of carbon contains:
NTA JEE Main 9th April 2019 Shift 1 - Question 39
The correct IUPAC name of the following compound is: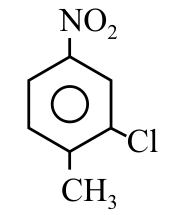
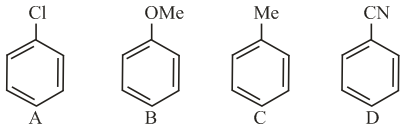
NTA JEE Main 9th April 2019 Shift 1 - Question 40
The increasing order of reactivity of the following compounds towards aromatic electrophilic substitution reaction is: