NTA JEE Main 9th April 2017 Online
For the following questions answer them individually
NTA JEE Main 9th April 2017 Online - Question 41
50 mL of 0.2 M ammonia solution is treated with 25 mL of 0.2 M HCl. If pK$$_b$$ of ammonia solution is 4.75, the pH of the mixture will be:
NTA JEE Main 9th April 2017 Online - Question 42
Which one of the following is an oxide?
NTA JEE Main 9th April 2017 Online - Question 43
Which of the following compounds is most reactive to an aqueous solution of sodium carbonate?
NTA JEE Main 9th April 2017 Online - Question 44
In the following structure, the double bonds are marked as I, II, III and IV.
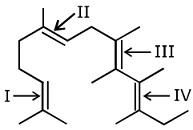
Geometrical isomerism is not possible at the site(s):
NTA JEE Main 9th April 2017 Online - Question 45
The major product of the following reaction is:
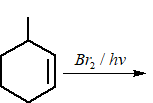
Methylcyclohexane reacts with Br$$_2$$/h$$\nu$$
NTA JEE Main 9th April 2017 Online - Question 46
Which of the following is a set of greenhouse gases?
NTA JEE Main 9th April 2017 Online - Question 47
A solution is prepared by mixing 8.5 g of CH$$_2$$Cl$$_2$$ and 11.95 g of CHCl$$_3$$. If vapour pressure of CH$$_2$$Cl$$_2$$ and CHCl$$_3$$ at 298K are 415 and 200 mm Hg respectively, the mole fraction of CHCl$$_3$$ in vapour form is: (Molar mass of Cl = 35.5 g mol$$^{-1}$$)
NTA JEE Main 9th April 2017 Online - Question 48
Which of the following ions does not liberate hydrogen gas on reaction with dilute acids?
NTA JEE Main 9th April 2017 Online - Question 49
To find the standard potential of M$$^{3+}$$/M electrode, the following cell is constituted:
Pt | M/M$$^{3+}$$ (0.001 mol L$$^{-1}$$) / Ag$$^+$$ (0.01 mol L$$^{-1}$$) / Ag
The emf of the cell is found to be 0.421 volt at 298 K. The standard potential of half-reaction M$$^{3+}$$ + 3e$$^-$$ $$\rightarrow$$ M at 298 K will be:
(Given: $$E^\ominus_{Ag^+/Ag}$$ at 298 K = 0.80 volt)
NTA JEE Main 9th April 2017 Online - Question 50
The rate of a reaction quadruples when the temperature changes from 300 to 310 K. The activation energy of this reaction is:
(Assume Activation energy and pre-exponential factor are independent of temperature;
ln(2) = 0.693; R = 8.314 J mol$$^{-1}$$ K$$^{-1}$$)


