NTA JEE Main 9th April 2017 Online
For the following questions answer them individually
NTA JEE Main 9th April 2017 Online - Question 31
What quantity (in mL) of a 45% acid solution of a mono-protic strong acid must be mixed with a 20% solution of the same acid to produce 800 mL of a 29.875% acid solution?
NTA JEE Main 9th April 2017 Online - Question 32
The electron in the hydrogen atom undergoes transition from higher orbitals to orbital of radius 211.6 pm. This transition is associated with
NTA JEE Main 9th April 2017 Online - Question 33
The electronic configuration with the highest ionization enthalpy is:
NTA JEE Main 9th April 2017 Online - Question 34
The group having triangular planar structure is:
NTA JEE Main 9th April 2017 Online - Question 35
The increasing order of the boiling points for the following compounds is:
(I) C$$_2$$H$$_5$$OH (II) C$$_2$$H$$_5$$Cl (III) C$$_2$$H$$_5$$CH$$_3$$ (IV) C$$_2$$H$$_5$$OCH$$_3$$
NTA JEE Main 9th April 2017 Online - Question 36
Among the following compounds, the compound which shows the highest dipole-moment is
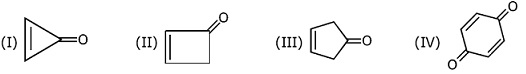
NTA JEE Main 9th April 2017 Online - Question 37
At 300 K, the density of a certain gaseous molecule at 2 bar is double to that of dinitrogen (N$$_2$$) at 4 bar. The molar mass of the gaseous molecule is
NTA JEE Main 9th April 2017 Online - Question 38
An ideal gas undergoes isothermal expansion at constant pressure. During the process:
NTA JEE Main 9th April 2017 Online - Question 39
A gas undergoes change from state A to state B. In this process, the heat absorbed and work done by the gas is 5 J and 8 J, respectively. Now gas is brought back to A by another process during which 3 J of heat is evolved. In this reverse process of B to A.
NTA JEE Main 9th April 2017 Online - Question 40
The following reaction occurs in the Blast Furnace where iron ore is reduced to iron metal:
Fe$$_2$$O$$_3$$(s) + 3CO(g) $$\rightleftharpoons$$ 2Fe(l) + 3CO$$_2$$(g)
Using the Le Chatelier's principle, predict which one of the following will not disturb the equilibrium?


