NTA JEE Main 8th January 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 8th January 2020 Shift 2 - Question 41
A metal (A) on heating in nitrogen gas gives compound B. B on treatment with H$$_2$$O gives a colourless gas which when passed through CuSO$$_4$$ solution gives a dark blue-violet coloured solution. A and B respectively, are:
NTA JEE Main 8th January 2020 Shift 2 - Question 42
The major product [B] in the following sequence of reactions is:
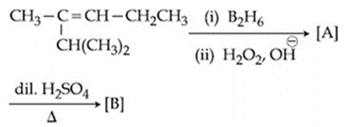
NTA JEE Main 8th January 2020 Shift 2 - Question 43
The correct order of the calculated spin-only magnetic moments of complexes (A) to (D) is:
(A) Ni(CO)$$_4$$
(B) [Ni(H$$_2$$O)$$_6$$]Cl$$_2$$
(C) Na$$_2$$[Ni(CN)$$_4$$]
(D) PdCl$$_2$$(PPh$$_3$$)$$_2$$
NTA JEE Main 8th January 2020 Shift 2 - Question 44
Two monomers in maltose are:
NTA JEE Main 8th January 2020 Shift 2 - Question 45
For the following Assertion and Reason, the correct option is:
Assertion: For hydrogenation reactions, the catalytic activity increases from Group 5 to Group 11 metals with maximum activity shown by Group 7-9 elements.
Reason: The reactants are most strongly adsorbed on group 7-9 elements.
NTA JEE Main 8th January 2020 Shift 2 - Question 46
NaClO$$_3$$ is used, even in spacecrafts, to produce O$$_2$$. The daily consumption of pure O$$_2$$ by a person in 492 L at 1 atm, 300K. How much amount of NaClO$$_3$$, in grams, is required to produce O$$_2$$ for the daily consumption of a person at 1 atm, 300K?
NaClO$$_3$$(s) + Fe(s) $$\rightarrow$$ O$$_2$$(g) + NaCl(s) + FeO(s)
R = 0.082 L atm mol$$^{-1}$$ K$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Question 47
At constant volume, 4 mol of an ideal gas when heated from 300K to 500K changes its internal energy by 5000J. The molar heat capacity at constant volume is
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Question 48
In the following sequence of reactions the maximum number of atoms present in molecule 'C' in one plane is __________.
A $$\xrightarrow[Cu tube]{\text{Red hot }}$$ B $$\xrightarrow[\text{Anhydrous AlCl}_3]{CH_3Cl(1 eq)}$$ C
(A is a lowest molecular weight alkyne)
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Question 49
For an electrochemical cell Sn(s)|Sn$$^{2+}$$(aq, 1M)||Pb$$^{2+}$$(aq, 1M)|Pb(s) the ratio $$\frac{[Sn^{2+}]}{[Pb^{2+}]}$$ when this cell attains equilibrium is __________.
(Given: E$$^0_{Sn^{2+}|Sn} = -0.14$$ V, E$$^0_{Pb^{2+}|Pb} = -0.13$$ V, $$\frac{2.303RT}{F} = 0.06$$)
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Question 50
Complexes (ML$$_5$$) of metals Ni and Fe have ideal square pyramidal and trigonal bipyramidal geometries, respectively. The sum of the 90$$^\circ$$, 120$$^\circ$$ and 180$$^\circ$$ L-M-L angles in the two complexes is
789
456
123
0.-
Clear All


