NTA JEE Main 8th January 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 8th January 2020 Shift 2 - Question 31
Among the reactions (a) - (d), the reaction(s) that does/do not occur in the blast furnace during the extraction of iron is/are:
(a) CaO + SiO$$_2$$ $$\rightarrow$$ CaSiO$$_3$$
(b) 3Fe$$_2$$O$$_3$$ + CO $$\rightarrow$$ 2Fe$$_3$$O$$_4$$ + CO$$_2$$
(c) FeO + SiO$$_2$$ $$\rightarrow$$ FeSiO$$_3$$
(d) FeO $$\rightarrow$$ Fe + $$\frac{1}{2}$$O$$_2$$
NTA JEE Main 8th January 2020 Shift 2 - Question 32
The radius of the second Bohr orbit, in terms of the Bohr radius, $$a_0$$, in Li$$^{2+}$$ is:
NTA JEE Main 8th January 2020 Shift 2 - Question 33
Kjeldahl's method cannot be used to estimate nitrogen for which of the following compounds?
NTA JEE Main 8th January 2020 Shift 2 - Question 34
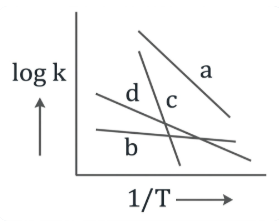
Consider the following plots of rate constant versus $$\frac{1}{T}$$ for four different reactions. Which of the following orders is correct for the activation energies of these reactions?
NTA JEE Main 8th January 2020 Shift 2 - Question 35
An unsaturated hydrocarbon X absorbs two hydrogen molecules on catalytic hydrogenation, and also gives following reaction:
X $$\xrightarrow[\frac{(ii) Zn}{H_2O}]{(i) O_3}$$ A $$\xrightarrow{[Ag(NH_3)_2]^+}$$ B (3-oxo-hexanedicarboxylic acid). X will be:
NTA JEE Main 8th January 2020 Shift 2 - Question 36
Which of the following compounds is likely to show both Frenkel and Schottky defects in its crystalline form?
NTA JEE Main 8th January 2020 Shift 2 - Question 37
Among (a) - (d), the complexes that can show geometrical isomerism are:
(a) [Pt(NH$$_3$$)$$_3$$Cl]$$^+$$
(b) [Pt(NH$$_3$$)Cl$$_5$$]$$^-$$
(c) [Pt(NH$$_3$$)$$_2$$Cl(NO$$_2$$)]
(d) [Pt(NH$$_3$$)$$_4$$ClBr]$$^{2+}$$
NTA JEE Main 8th January 2020 Shift 2 - Question 38
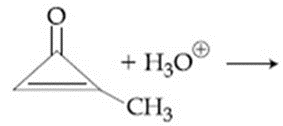
The major product in the following reaction is:
NTA JEE Main 8th January 2020 Shift 2 - Question 39
White phosphorus on reaction with concentrated NaOH solution in an inert atmosphere of CO$$_2$$ gives phosphine and compound (X). (X) on acidification with HCl gives compound (Y). The basicity of compound (Y) is:
NTA JEE Main 8th January 2020 Shift 2 - Question 40
Hydrogen has three isotopes (A), (B) and (C). If the number of neutron(s) in (A), (B) and (C) respectively, are (x), (y) and (z), the sum of (x), (y) and (z) is


