NTA JEE Main 8th April 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 8th April 2019 Shift 2 - Question 41
Polysubstitution is a major drawback in:
NTA JEE Main 8th April 2019 Shift 2 - Question 42
Which one of the following alkenes when treated with HCl yields majorly an anti Markovnikov product?
NTA JEE Main 8th April 2019 Shift 2 - Question 43
The maximum prescribed concentration of copper in drinking water is:
NTA JEE Main 8th April 2019 Shift 2 - Question 44
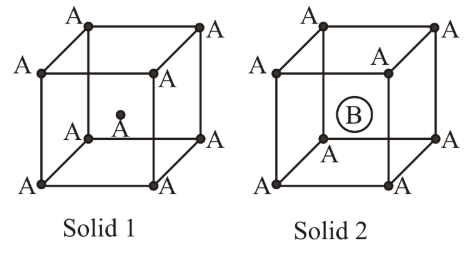
Consider the bcc unit cells of the solids 1 and 2 with the position of atoms as shown below. The radius of atom B is twice that of atom A. The unit cell edge length is 50% more in solid 2 than in 1. What is the approximate packing efficiency in solid 2?
NTA JEE Main 8th April 2019 Shift 2 - Question 45
For the solution of the gases w, x, y and z in water at 298 K, the Henry's law constants (K$$_H$$) are 0.5, 2, 35 and 40 kbar, respectively. The correct plot for the given data is:
NTA JEE Main 8th April 2019 Shift 2 - Question 46
Calculate the standard cell potential (in V) of the cell in which the following reaction takes place:
Fe$$^{2+}$$(aq) + Ag$$^+$$(aq) $$\rightarrow$$ Fe$$^{3+}$$(aq) + Ag(s)
Given that $$E^{0}_{Ag^+/Ag} = x$$ V, $$E^{0}_{Fe^{2+}/Fe} = y$$ V, $$E^{0}_{Fe^{3+}/Fe} = z$$ V
NTA JEE Main 8th April 2019 Shift 2 - Question 47
For a reaction scheme A $$\xrightarrow{k_1}$$ B $$\xrightarrow{k_2}$$ C, if the net rate of formation of B is set to be zero then the concentration of B is given by:
NTA JEE Main 8th April 2019 Shift 2 - Question 48
The Mond process is used for the:
NTA JEE Main 8th April 2019 Shift 2 - Question 49
The ion that has sp$$^3$$d$$^2$$ hybridization for the central atom is:
NTA JEE Main 8th April 2019 Shift 2 - Question 50
The correct statement about ICl$$_5$$ and ICl$$_4^{-}$$ is:


