NTA JEE Main 8 April 2018 Offline
For the following questions answer them individually
NTA JEE Main 8 April 2018 Offline - Question 31
The ratio of mass percent of C and H of an organic compound ($$C_XH_YO_Z$$) is 6 : 1. If one molecule of the above compound ($$C_XH_YO_Z$$) contains half as much oxygen as required to burn one molecule of compound $$C_XH_Y$$ completely to $$CO_2$$ and $$H_2O$$. The empirical formula of the compound $$C_XH_YO_Z$$ is:
NTA JEE Main 8 April 2018 Offline - Question 32
According to molecular orbital theory, which of the following molecule will not be available?
NTA JEE Main 8 April 2018 Offline - Question 33
Which of the following compounds contain(s) no covalent bond(s)?
KCl, PH$$_3$$, O$$_2$$, B$$_2$$H$$_6$$, H$$_2$$SO$$_4$$
NTA JEE Main 8 April 2018 Offline - Question 34
Total number of lone pair of electrons in I$$_3^-$$ ion is:
NTA JEE Main 8 April 2018 Offline - Question 35
The combustion of benzene (l) gives CO$$_2$$(g) and H$$_2$$O(l). Given that heat of combustion of benzene at constant volume is $$-3263.9$$ kJ mol$$^{-1}$$ at 25$$^\circ$$C; the heat of combustion (in kJ mol$$^{-1}$$) of benzene at constant pressure will be:
(R = 8.314 JK$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 8 April 2018 Offline - Question 36
Which of the following lines correctly show the temperature dependence of equilibrium constant K, for an exothermic reaction?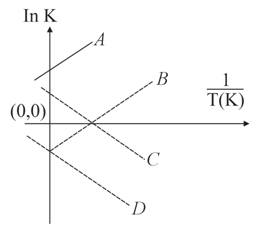
NTA JEE Main 8 April 2018 Offline - Question 37
An aqueous solution contains 0.10 M H$$_2$$S and 0.20 M HCl. If the equilibrium constant for the formation of HS$$^-$$ from H$$_2$$S is $$1.0 \times 10^{-7}$$ and that of S$$^{2-}$$ from HS$$^-$$ ions is $$1.2 \times 10^{-13}$$, then the concentration of S$$^{2-}$$ ions in the aqueous solution is:
NTA JEE Main 8 April 2018 Offline - Question 38
An aqueous solution contains an unknown concentration of Ba$$^{2+}$$. When 50 mL of a 1 M solution of Na$$_2$$SO$$_4$$ is added, BaSO$$_4$$ just begins to precipitate. The final volume is 500 mL. The solubility product of BaSO$$_4$$ is $$1 \times 10^{-10}$$. What is the original concentration of Ba$$^{2+}$$?
NTA JEE Main 8 April 2018 Offline - Question 39
Which of the following are Lewis acids?
NTA JEE Main 8 April 2018 Offline - Question 40
Which of the following salts is the most basic in aqueous solution?


