NTA JEE Main 7th January 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 7th January 2020 Shift 2 - Question 41
For the following reactions
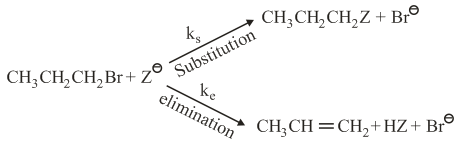
where:
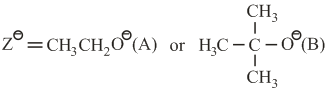
k$$_s$$ and k$$_e$$ are respectively the rate constants for substitution and elimination, and $$\mu = \frac{k_s}{k_e}$$, the correct option is
NTA JEE Main 7th January 2020 Shift 2 - Question 42
Consider the following reactions:
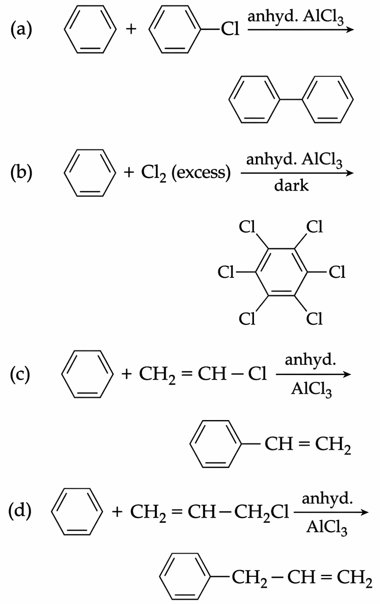
Which of these reactions are possible?
NTA JEE Main 7th January 2020 Shift 2 - Question 43
In the following reaction sequence, structures of A and B are, respectively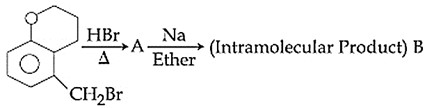
Cyclohexanone-CH$$_2$$Br $$\xrightarrow{\text{HBr}}$$ A $$\xrightarrow{\text{Na/Ether}}$$ (Intramolecular Product) B
NTA JEE Main 7th January 2020 Shift 2 - Question 44
In the following reaction sequence,
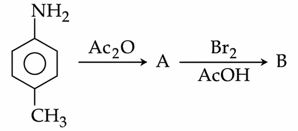
the major product B is
NTA JEE Main 7th January 2020 Shift 2 - Question 45
Which of the following statements is correct?
NTA JEE Main 7th January 2020 Shift 2 - Question 46
The standard heat of formation $$(\Delta_f H_{298}^0)$$ of ethane (in kJ/mol), if the heat of combustion of ethane, hydrogen and graphite are $$-1560$$, $$-393.5$$ and $$-286$$ kJ/mol, respectively is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Question 47
3 g of acetic acid is added to 250 mL of 0.1 M HCl and the solution made up to 500 mL. To 20 mL of this solution $$\frac{1}{2}$$ mL of 5M NaOH is added. The pH of the solution is
[Given: pKa of acetic acid = 4.75, molar mass of acetic acid 60 g/mol, log 3 = 0.4771, Neglect any changes in volume]
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Question 48
Consider the following reactions:
NaCl + K$$_2$$Cr$$_2$$O$$_7$$ + H$$_2$$SO$$_4$$ (Conc.) $$\rightarrow$$ (A) + side products
(A) + NaOH $$\rightarrow$$ (B) + side products
(B) + H$$_2$$SO$$_4$$ + H$$_2$$O$$_2$$ (dilute) $$\rightarrow$$ (C) + side products
The sum of the total number of atoms in one molecule each of (A), (B) and (C) is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Question 49
The flocculation value of HCl for arsenic sulphide sol is 30 mmolL$$^{-1}$$. If H$$_2$$SO$$_4$$ is used for the flocculation of arsenic sulphide, the amount, in grams, of H$$_2$$SO$$_4$$ in 250 ml required for the above purpose is (molecular mass of H$$_2$$SO$$_4$$ = 98 g/mol)
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Question 50
The number of sp$$^2$$ hybridised carbons present in "Aspartame" is
789
456
123
0.-
Clear All


